Christiane SimioniI; Edward Araujo JúniorII; Wellington P. MartinsIII; Liliam Cristine RoloIV; Luciane Alves da RochaV; Luciano Marcondes Machado NardozzaVI; Antonio Fernandes MoronVII
DOI: 10.5935/1678-9741.20120058
CRL: Crown-rump length
C O: Cardiac output
CHD: Congenital heart disease
SD: Standard deviation
EF: Ejection fraction
EFW: Estimation of fetal weight
FHR: Fetal heart rate
EF: Ejection fraction
GA: Gestational age in weeks
ROI: Region of Interest
STIC: Spatio-temporal image correlation
UNIFESP: Federal University of Sao Paulo
US 2D: Bi-dimensional ultrasonound
US 3D: Three-dimensional ultrasound
VOCAL: Virtual organ computer-aided analysis
SV: Systolic volume
INTRODUCTION
Congenital heart disease (CHD) accounts for significant portion of the rate of perinatal morbidity and mortality by both anatomical and functional cardiac defects. The DCC is the most common major malformations at birth [1,2], with a prevalence of 0.6 to 5% of live births [3]. Despite great efforts and technological advancement of two-dimensional echocardiography in the past two decades, the accuracy in detecting congenital heart disease in pre-natal is between 31% to 96% [4,5].
Despite the two-dimensional ultrasound (2D U.S.) is used for the study of cardiac anatomy with good accuracy for the analysis of fetal cardiac function by means of this diagnostic modality is still limited [6]. The analysis of the stroke volume (SV) and ejection fraction (EF) uses the formula Teichholz, which can be applied to wells of any size, assuming the ventricle has an elliptical shape, or using only a constant (X x Y x Z x 0.52) [7]. The Simpson's method is also employed for this purpose, but even more complex by dividing the ventricle in multi-cylinder, calculating the EP to each medium and maintaining the total of the fractions isolated [8].
The three-dimensional ultrasonography (3D U.S.) allows a more accurate volumetric evaluation that 2D ultrasound, especially for objects of irregular shapes, it allows the design of its outer surface [9], which may be of potential use for evaluation of fetal cardiac function. The Spatio-Temporal Image Correlation (STIC) is a software that allows a volumetric acquisition of the fetal heart with its vascular connections, and the images can be evaluated both on the multiplanar mode and surface (rendered). The images may be evaluated both a static and moving (4D) by means of a Cineloop sequence that simulates a complete cardiac cycle. The advantages of STIC to evaluate fetal heart are less dependent on operator's experience in obtaining diagnostic plans, shorter examination carried out with analysis of volumes in the absence of the patient, ability to assess structures by rendering mode to study their morphology and function [10-12]. With respect to cardiac evaluation at STIC, first study was performed by Messing et al. [13], who evaluated the volume of the ventricular chambers in both systole and diastole in 100 fetuses using methods Virtual Organ Computer-aided Analysis (VOCAL) associated with the inversion mode. Later, other authors also evaluated fetal cardiac function and Molina et al. [14] who used the STIC associated only with VOCAL, Uittenbogaard et al. [15] who used the STIC and 3D slice method and, more recently, Simioni et al. [16] and Hamill et al. [17], using only the VOCAL associated with STIC. However, no studies comparing cardiac function by means of 3D U.S. with STIC in relation to gender. It is known that in adults, by means of 3D echocardiography, women have a higher volume / ventricular mass than men of similar age [18].
The objective of this study was to compare cardiac output (CO) and EF heart male and female fetuses by 3D ultrasound using STIC and VOCAL software in order to try to determine if cardiac function is already dependent on gender antenatal period.
METHODS
We conducted a cross-sectional study, from May 2009 to July 2011, with healthy pregnant women between 20-34 weeks and 6 days. This study was approved by the Ethics Committee in Research of the Federal University of Sao Paulo (UNIFESP) No 0234/09, and the patients who consented to participate voluntarily signed a consent form.
Inclusion criteria were: 1) singleton pregnancy with live fetus, 2) gestational age determined by last menstrual period and confirmed by first trimester ultrasound, using as parameter the crown-rump length (CRL), with a difference less than 5 days.
Exclusion criteria were: 1) the fetal position with the dorsal anterior (between 11 and 1h), 2) excessive fetal movements, 3) severe attenuation of the sound beam (obesity, abdominal scars), 4) fetal malformation detected on ultrasound, 5) estimated fetal weight below the 10th percentile or above the 90th percentile, according to Hadlock et al. [19], 6) amniotic fluid index below the 5th percentile or above the 95th percentile, according to Moore & Cayle [20], 7) maternal chronic diseases that could interfere with fetal growth (chronic hypertension, diabetes mellitus and collagen) ; 8) cases in which it was not possible to identify the fetal external genitalia.
All patients were selected randomly, and these are coming from the Division of Prenatal Physiology, Department of Obstetrics, UNIFESP, or Basic Health Units of the metropolitan region of São Paulo (Brazil). Patients were evaluated only once, no data were obtained postnatally.
The following variables were evaluated pregnant women included: age, number of previous pregnancies and deliveries, gestational age, fetal weight, fetal heart rate (FHR), fetal gender, DC right ventricular and left ventricular EF and combined right and left. Age, number of previous pregnancies and deliveries of the pregnant women were obtained by questionnaires. The other variables were evaluated by ultrasonography.
The tests were conducted at Centro Paulista de Medicina Fetal (CPMF) and the Division of Three-Dimensional Ultrasound, Department of Obstetrics, UNIFESP, which were accomplished by only two investigators (CS and LLC), both with three years experience in 3D ultrasound in Obstetrics. All examinations were performed on branded handsets Voluson 730 Expert (General Electric Healthcare, Zipf, Austria) using a multifrequency convex volumetric transducer (RAB 4-8L). Initially, the observer, by means of 2D U.S., carried out the measurements of biparietal diameter, abdominal circumference and femur length to determine the estimated fetal weight [21]. The determination of the genus fetal was performed using the 2D U.S. [22]. The fetal heart rate (FHR) was determined by pulsed Doppler ultrasonography of the mitral valve. For the acquisition of the fetal heart volume, initially there was a two-dimensional evaluation in order to obtain an axial level of the four heart chambers. Then, with the fetus asking the home and pregnant breath for a few seconds, there was a 3D scan. Therefore, if the key-activated STIC, so that the region of interest (ROI) encompassing only the heart with their vascular connections. The sweep angle and speed of acquisition was determined by the examiner. The angle ranged from 20º to 25º in the first quarter, and 25º to 30º in the third quarter, while the acquisition time from 10 to 12.5 seconds. After the capture volume, it was found and the quality of that which is considered satisfactory, it was stored in the memory. After standardization of the position of the fetal heart Paladini proposed by [23], a single volume of each fetal heart was stored in the memory. Subsequently, volumes were recorded on compact discs (CDs) and transferred to a personal computer (PC).
The analyses were performed offline using the software 4D View version 9.1 (General Electric Healthcare). The axial plane of the multiplanar mode was selected as a reference. In order to evaluate the dynamics of the mitral and tricuspid valves, gradually reduce the speed of cinellop to obtain exact opening and closing of valves. The maximum diameter of the ventricle to the mitral and tricuspid valves determined the closed end of the ventricular diastole, whereas the smaller diameter end determined the ventricular systole. Then the key is activated VOCAL with a rotation angle of 30º (setting six consecutive planes). Thus, the reference point was moved to the center of the ventricle, and the axial plane rotated around the axis "y", so the cardiac apex is available within 12 hours. It was performed manually six planes defining the left ventricle after which the device provided the reconstructed image with the volume of the ventricular cavity. The same process was repeated for the right ventricle. Finally, there were obtained the volumes of the right and left ventricles, either at the end of systole and in the end of diastole. The stroke volume of each ventricle was determined by the formula: stroke volume = (ventricular volume at end diastole) - (ventricular volume at end systole). The DC each ventricle was determined by the equation: DC = (stroke volume) * (FCF). The combined DC was determined by adding the values obtained for the DC of the right ventricle and left. The EF of each ventricle was determined by the formula: EF = (stroke volume) / (ventricular volume at end diastole).
Statistical analysis was performed using the programs Excel 2007 (Microsoft Corp., Redmond, WA, USA), PASW (version 18.0, SPSS Inc., Chicago, IL, USA) and GraphPad (version 5.0, GraphPad Software, San Diego, CA, USA). The normal distribution was assessed using the Kolmogorov-Smirnov test. We compared maternal age gestational age, fetal weight and FCF between the fetus pregnancies with female and male by means of the Student's t-test. Number of pregnancies and parity between groups was compared using the Mann-Whitney test. The DC (right, , left and combined) and EF (right and left) were compared between groups by unpaired t-test and also by ANCOVA, using gestational age and / or estimated fetal weight as covariates. Since we did not notice a difference in the evaluated parameters between fetuses were male and female, were determined curves of normal (5th percentile, 50 and 95) by the mean and standard deviation for the estimated gestational age [24], using data from all fetuses.
RESULTS
We initially evaluated 290 pregnant women; however, 34 were excluded due to unfavorable fetal position, 5 per estimated fetal weight below the 10th percentile for gestational age and 35 for failing to identify fetal sex. So for the final statistical analysis we included 216 fetuses, with 108 of each sex.
There was no significant difference between the fetuses of pregnant women with male and female regarding maternal age, number of previous pregnancies and deliveries, gestational age, fetal weight and fetal heart rate (Table 1).
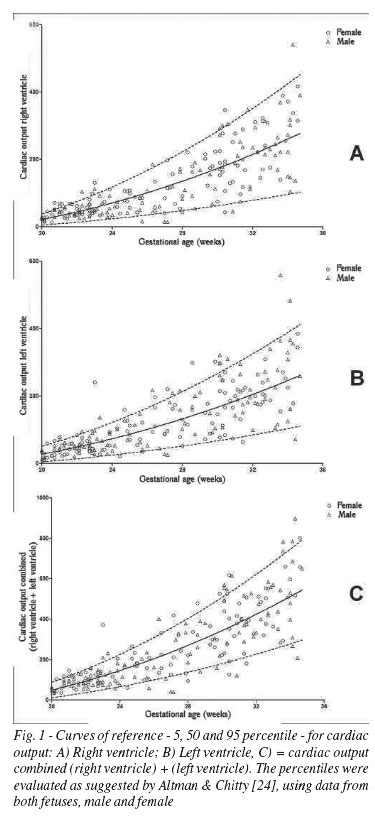
No significant differences when comparing the CD and EF between the fetuses were male and female, both the analysis of initial data, even when adjusting for gestational age, estimated fetal weight, or both (Table 2). Thus, the percentiles for the CD and EF were calculated for gestational ages between 26 and 35 weeks, using data both from male fetuses as female (Tables 3 and 4, Figures 1 and 2).


DISCUSSION
In this study, we evaluated fetal cardiac function by means of 3D U.S., using the softwares STIC and VOCAL. We used the limit range of 20 to 34 weeks, because during this period the visualization of cardiac chambers is easier, providing better heart volume. In addition, below 20 weeks postnatal fetal viability is not possible, making it unnecessary to evaluation of their cardiac function. Used as standards for acquiring volumetric cutting four cardiac chambers, where possible position of the back in 6 hours, a sweep angle ranging from 20 º to 30 º according to the gestational age, as long as possible to acquire, in addition to exclusion of cases with back between 11 and 1am, as proposed by Gonçalves et al. [25].
This study aimed to evaluate possible differences in cardiac function according to fetal gender. However, we could not demonstrate significant differences for both the DC and for the FE, both adjusted for gestational age and the estimated fetal weight. In a study using 3D transthoracic echocardiography, the volume / left ventricular mass was significantly higher in women than in men of similar age. Another cause could be the largest pulmonary resistance of women relative to men, as a possible consequence of increased left ventricular EF. A previous study by 2D echocardiography has shown that heart failure with normal EF is often more common in women than in men of similar age [26]. Possibly, external factors acting in the postnatal period may justify the difference in cardiac function in relation to gender, in adults of the same age.
In this study, 290 pregnant women were initially evaluated, 34 were excluded due to poor quality of packages corresponding to a rate of 11.7% loss. In a recent study by Schoonderwaldt et al. [27], 84 women were initially evaluated between 20-34 weeks, however, excluded volume 54, corresponding to a rate of 64% loss. This high rate of loss due to low quality of cardiac volumes is due to inclusion of the previous position back as an exclusion criterion and the small number of cases evaluated in comparison to the study conducted by us.
In our study, as we have not identified differences in cardiac function in male and female fetuses, we determined reference values only for both the DC (right, left and combined) and for EF (right and left). We opted for the determination of reference values from 26 weeks, taking into account fetal viability. In relation to the previous study by Molina et al. [14], who evaluated 100 fetuses of both sexes, between 12 and 34 weeks, we observed that their values were overestimated compared to ours. We believe that one possible reason is the inclusion in the endocardium volume calculation, while in this study only the area delimited anechoic (blood) present in the cardiac chambers, the same as in previous studies by our group [16.28] . In comparison with longitudinal study carried out by Uittenbogaard et al. [15] who used the technique 3D slice, we observed that in the range 26 to 30 weeks, our values were also underestimated the likely cause is possibly also the inclusion of the endocardium in the volumetric calculations performed in that study. In a recent study by Hamill et al. [17], who evaluated 180 cardiac volumes by STIC and VOCAL, we found that DC increased with gestational age and did not differ between the right and left ventricles, whereas EF decreased with gestational age and was higher in the left ventricle . These results are in agreement with those obtained in our study, with 216 fetuses of both sexes.
This study did not evaluate the reproducibility of the method STIC and VOCAL, because it has been proven in a previous study conducted by our group [16], as well as other studies [14,27,29].
Limitations of this study relate primarily to the capture volume, which requires a fetal position right (back in 6 hours) or semi-ideal (back between 3 and 9), poor fetal movements and apnea in pregnant women, which sometimes makes testing extremely time consuming. Furthermore, the post-processing of images, with calculations of ventricular volumes at the end of systole and diastole, to obtain the CD and EF consumes a long time (10-12 minutes), making their use in clinical practice. Another limitation relates to biotype and the presence of maternal abdominal scars generating acoustic shadows difficult and sometimes impossible to capture a stroke volume to a satisfactory quality. The same limitations have already been described in a previous study carried out by Hamill et al. [29].
CONCLUSION
In summary, this is the first study that sought to assess differences in the intrauterine fetal cardiac function in relation to gender by US 3D. The lack of statistical difference observed in the CD and EF of male and female fetuses implies that external factors postnatal are responsible for these observed differences in adult life for people of different sex and same age. Further studies evaluating the cardiac function by means of 3D U.S. in childhood and adolescence are necessary to confirm our assumption.
REFERENCES
1. Silva MEM, Feuser MR, Silva MP, Uhlig S, Parazzi PLF, Rosa GJ, et al. Cirurgia cardíaca pediátrica: o que esperar da intervenção fisioterapêutica? Rev Bras Cir Cardiovasc. 2011;26(2):264-72. [MedLine] View article
2. Hoffman JI. Incidence of congenital heart disease: I. Postnatal incidence. Pediatr Cardiol. 1995;16(3):103-13. [MedLine]
3. Grandjean H, Larroque D, Levi S. The performance of routine ultrasonographic screening of pregnancies in the Eurofetus Study. Am J Obstet Gynecol. 1999;181(2):446-54. [MedLine]
4. Strumpflen I, Strumpflen A, Wimmer M, Bernaschek G. Effect of detailed fetal echocardiography as part of routine prenatal ultrasonographic screening on detection of congenital heart disease. Lancet. 1996;348(9031):854-7. [MedLine]
5. Berghella V, Pagotto L, Kaufman M, Huhta JC, Wapner RJ. Accuracy of prenatal diagnosis of congenital heart defects. Fetal Diagn Ther. 2001;16(6):407-12. [MedLine]
6. Esh-Broder E, Ushakov FB, Imbar T, Yagel S. Application of free-hand three-dimensional echocardiography in the evaluation of fetal cardiac ejection fraction: a preliminary study. Ultrasound Obstet Gynecol. 2004;23(6):546-51. [MedLine]
7. Schmidt KG, Silverman NH, Hoffman JI. Determination of ventricular volumes in human fetal hearts by two-dimensional echocardiography. Am J Cardiol. 1995;76(17):1313-16. [MedLine]
8. Meyer-Wittkopf M, Cole A, Cooper SG, Schmidt S, Sholler GF. Three-dimensional quantitative echocardiographic assessment of ventricular volume in healthy human fetuses and in fetuses with congenital heart disease. J Ultrasound Med. 2001;20(4):317-27. [MedLine]
9. Riccabona M, Nelson TR, Pretorius DH. Three-dimensional ultrasound: accuracy of distance and volume measurements. Ultrasound Obstet Gynecol. 1996;7(6):429-34. [MedLine]
10. Gonçalves LF, Lee W, Chaiworapongsa T, Espinoza J, Schoen ML, Falkensammer P, et al. Four-dimensional ultrasonography of the fetal heart with spatiotemporal image correlation. Am J Obstet Gynecol. 2003;189:1792-802. [MedLine]
11. DeVore GR, Falkensammer P, Sklansky MS, Platt LD. Spatiotemporal image correlation (STIC): new technology for evaluation of the fetal heart. Ultrasound Obstet Gynecol. 2003;22(4):380-7. [MedLine]
12. Vinãls F, Poblete P, Giuliano A. Spatio-temporal image correlation (STIC): a new tool for the prenatal screening of congenital heart defects. Ultrasound Obstet Gynecol. 2003;22(4):388-94. [MedLine]
13. Messing B, Cohen SM, Valsky DV, Rosenak D, Hochner-Celnikier D, Savchev S, et al. Fetal cardiac ventricle volumetry in the second half of gestation assessed by 4D ultrasound using STIC combined with inversion mode. Ultrasound Obstet Gynecol. 2007;30(2):142-51. [MedLine]
14. Molina FS, Faro C, Sotiriadis A, Dagklis T, Nicolaides KH. Heart stroke volume and cardiac output by four-dimensional ultrasound in normal fetuses. Ultrasound Obstet Gynecol. 2008;32(2):181-7. [MedLine]
15. Uittenbogaard LB, Haak MC, Spreeuwenberg MD, van Vugt JM. Fetal cardiac function assessed with four-dimensional ultrasound imaging using spatiotemporal image correlation. Ultrasound Obstet Gynecol. 2009;33(3):272-81. [MedLine]
16. Simioni C, Nardozza LM, Araujo Júnior E, Rolo LC, Zamith M, Caetano AC, et al. Heart stroke volume, cardiac output, and ejection fraction in 265 normal fetus in the second half of gestation assessed by 4D ultrasound using spatio-temporal image correlation. J Matern Fetal Neonatal Med. 2011;24(9):1159-67. [MedLine]
17. Hamill N, Yeo L, Romero R, Hassan SS, Myers SA, Mittal P, et al. Fetal cardiac ventricular volume, cardiac output, and ejection fraction determined with 4-dimensional ultrasound using spatiotemporal image correlation and virtual organ computer-aided analysis. Am J Obstet Gynecol. 2011;205(1):76.e1-10.
18. Kaku K, Takeuchi M, Otani K, Sugeng L, Nakai H, Haruki N, et al. Age-and gender-dependency of the left ventricular geometry assessed with real-time three-dimensional transthoracic echocardiography. J Am Soc Echocardiogr. 2011;24(5):541-7. [MedLine]
19. Hadlock FP, Harrist RB, Martinez-Poyer J. In utero analysis of fetal growth: a sonographic weight standard. Radiology. 1991;181(1):129-33. [MedLine]
20. Moore TR, Cayle JE. The amniotic fluid index in normal human pregnancy. Am J Obstet Gynecol. 1990;162(5):1168-73. [MedLine]
21. Scholly TA, Sutphen JH, Hitchcock DA, Mackey SC, Langstaff LM. Sonographic determination of fetal gender. AJR Am J Roentgenol. 1980;135(6):1161-5. [MedLine]
22. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements--a prospective study. Am J Obstet Gynecol. 1985;151(3):333-7. [MedLine]
23. Paladini D. Standardization of on-screen fetal heart orientation prior to storage of spatio-temporal image correlation (STIC) volume datasets. Ultrasound Obstet Gynecol. 2007;29(6):605-11. [MedLine]
24. Altman DG, Chitty LS. Charts of fetal size: 1. Methodology. Br J Obstet Gynaecol. 1994;101(1):29-34. [MedLine]
25. Gonçalves LF, Lee W, Espinoza J, Romero R. Examination of the fetal heart by four-dimensional (4D) ultrasound with spatio-temporal image correlation (STIC). Ultrasound Obstet Gynecol. 2006;7(3):336-48.
26. Regitz-Zagrosek V, Brokat S, Tschope C. Role of gender in heart failure with normal left ventricular ejection fraction. Prog Cardiovasc Dis. 2007;49(4):241-51. [MedLine]
27. Schoonderwaldt EM, Groenenberg IA, Hop WC, Wladimiroff JW, Steergers EA. Reproducibility of echocardiography measurements of human fetal left ventricular volumes and ejection fractions using four-dimensional ultrasound with the spatio-temporal image correlation modality. Eur J Obstet Gynecol Reprod Biol. 2012;160(1):22-9. [MedLine]
28. Simioni C, Nardozza LM, Araujo Júnior E, Rolo LC, Terasaka OA, Zamith MM, et al. Fetal cardiac function assessed by spatio-temporal image correlation. Arch Gynecol Obstet. 2011;284(1):253-60. [MedLine]
29. Hamill N, Romero R, Hassan SS, Lee W, Myers SA, Mittal P, et al. Repeatability and reproducibility of fetal cardiac ventricular volume calculations using spatiotemporal image correlation and virtual organ computer-aided analysis. J Ultrasound Med. 2009;28(10):1301-11. [MedLine]
Article receive on Thursday, March 1, 2012
 All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license
All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license