Juán Carlos Pachón MateosI; José Carlos Pachón MateosII; Remy Nelson Albornoz VargasI; Enrique Indalécio Pachón MateosI; Khalil CosacI; Hugo Belloti LopesI; Fabrizio Achilles SoaresI; Amanda Guerra Moraes Rego SousaIII
DOI: 10.5935/1678-9741.20120055
AV: block Atrioventricular block
Complete AV block: Complete atrioventricular block
DECA: Departamento de Estimulação Cardíaca Artificial [Artificial Heart Stimulation Department]
SND: Sinus Node dysfunction
AF: Atrial Fibrillation
EF: Ejection Fraction
CAD: Coronary Artery Disease
NYHA: New York Heart Association
RV: Right Ventricle
SOBRAC: Sociedade Brasileira de Arritmias Cardíacas [Brazilian Society of Cardiac Arrhythmias]
LV: Left Ventricle
INTRODUCTION
The normal QRS duration is < 120 ms owing to the very rapid ventricular activation mediated by the His-Purkinje system and by the subendocardial branches of Purkinje. This organized activation also determines the location of the normal QRS axis to the left (between -30 and +90 degrees) and backwards, pointing to the left ventricle (LV) due to its electrical predominance. Apart from speed and synchronism, the normal course of the cardiac conduction favors the optimum maintenance of the intramyocardial tensions. These benefits are lost when the QRS widens, which results in significant damage of the cardiac function [1-3]. The wide QRS (mainly with morphology of left bundle branch block-LBBB) promotes ventricular desynchronization. Thus, while part of the cells is contracting the other part is still relaxing, blunting the increase in intraventricular pressure which creates a high preload in lately activated cells. Eventually, the desynchronized activation of the papillary muscles worsens or even provokes mitral regurgitation. These abnormalities result in significant loss of ventricular effectiveness, especially in cases with cardiomyopathy.
The conventional cardiac pacemaker with the implanted lead in the right ventricle (RV) apex promotes an important widening of the QRS which is similar in morphology and mechanical dyssynchrony to the one caused by LBBB [4-6]. This phenomenon is an important factor which promotes an unwanted ventricular remodeling [7-9]. Great progress has been achieved to prevent, correct or reduce the ventricular desynchronization, such as biventricular (BiV) pacing [10,11] or the bifocal right ventricular pacing [12,13] which depend on placement of one more ventricular lead. Septal pacing has been widely used in the search for a less deleterious right ventricular pacing, but preliminary data have possibly shown isolated cases with higher thresholds and shorter R waves.
Primary Endpoint
To compare apical and septal pacing during acute and chronic phases in the same patient. It aims to identify any significant differences in threshold, R wave and impedance which may reject or recommend any pacing point.
Secondary Endpoints
1. To compare the QRS durations originated by septal and apical pacing;
2. To check if the QRS axis is more or less physiological (between -30 degrees and +90 degrees) in septal pacing in relation to apical pacing;
3. To evaluate the stability of the septal lead and the risk of displacement.
METHODS
The features of the patients of this trial are summarized in Table 1.
The pacemakers indications were determined according to SOBRAC-DECA and American Heart Association [14] guidelines.
Implantation Technique
In each patient two leads were intravenously implanted, one in the RV apex, in the classical position of the conventional endocardial pacing, and another in high portions of the intraventricular septum next to His Bundle, called septal in this trial, searching for the best command and sensitivity parameters. The lead was placed in high, mid-septal or para-Hisian region, and that of narrower QRS was chosen. The endocardial injury current in endocavitary electrogram was registered and evaluated in both septal and apical implantations, searching for a good myocardial viability and for the best placement of the lead.
In order to access the high septum, it was used a manual modeling of a steel guide in two planes, so that it could be directed to the high portions with its tip turned backwards. This position was confirmed by radioscopy in left anterior oblique position (Figure 1), aiming to prevent unintended and unwanted implantation into the RV free wall.
It was used endocardial leads from three manufacturers with the following features: active fixation by screw-in, narrow diameter, short distance between the poles and similar impedances. Biotronik DR Philos II and Entovis were used because they were the only pacemakers allowing programming to a DVIR mode with an very short AV interval of 15 ms. These resources are indispensable for the RV bifocal pacing, since it allows virtually simultaneous activation of the two points being the high septum 15 ms prior to the RV apex. Additionally this mode of pacing enables a noninvasive and independent programming of the two points, at any stage of the follow-up.
Unipolar and bipolar parameters - threshold, R wave and impedance - in both positions were evaluated by Biotronik ICS-300 system during the implantation and six months later by telemetry.
The 12-lead electrocardiograms (EKG) were recorded by the TEB ECG PC computerized electrocardiograph.
During the follow-up all the patients were kept with RV bifocal pacing as this stimulation was the one with the narrower paced QRS.
The patients were observed for 60 days to check possible complications associated with the implantation.
Statistical Analysis and Informed Consent
Data were inserted on Excel-2010 spreadsheet and means, standard deviation, maximum and minimum values, confidence intervals as well as median were established. The continuous variables were evaluated by two-tailed paired t test. The differences with P value < 0.05 were considered significant. Informed consent was obtained from all patients for pacemaker and lead implantation and for all the measurements during the follow-up.
RESULTS
The pacing thresholds, R wave and impedances unipolar and bipolar, acute and chronic at the sixth month of followup are shown in Table 2.
The means of septal and apical values unipolar and bipolar were very similar without significant statistical difference (P > 0.09) except when comparing unipolar thresholds in chronic phase, in which the mean of unipolar ones was slightly lower in septal pacing than in apical pacing, 0.5Vx 0.72V, (P=0.02).
Table 3 shows the comparisons of QRS duration obtained from the 12-lead ECG in the chronic phase, in apical and septal pacing. The mean of QRS duration obtained by septal pacing was clearly lower than the mean of QRS duration of apical pacing with highly significant difference (P<0.01).
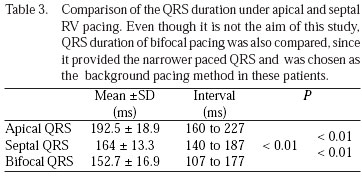
Incidentally, were observed that the mean of QRS duration obtained by RV bifocal pacing (being septum 15 ms before apex) was even lower.
In terms of the QRS axis there was a clear tendency of keeping the axis inside the normal limits under septal pacing, while the apical pacing caused complete non-physiological QRS axis, (Table 4). This difference was statistically very significant.
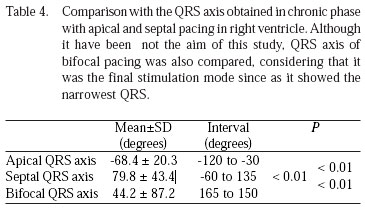
Although being not the objective of the study, bifocal QRS axis were measured to determine the best method to stimulate the enrolled patients.
Complications
There was one lead displacement from the septal position and one hematoma caused by the oral anticoagulant use as these patients presented permanent atrial fibrillation. The lead was replaced with one with longer screw-in system and the hematoma was easily solved with surgical repair before the hospital discharge.
DISCUSSION
Many patients who suffer from heart failure (HF) due to dilated cardiomyopathy (DCM) show bradyarrhythmia with cardiac pacemaker indication. However, classical ventricular pacing of the RV apex with wide QRS causes systolic and diastolic function impairment, besides mitral regurgitation [7]. Consequently, although the bradyarrhythmia be solved, the classical (apical) pacing worsens or even triggers HF. The functional impairment is even greater in patients with cardiomyopathy who are long-term pacemaker dependent and usually show widen QRS such as LBBB, reaching even more than 250 ms. The same unwanted hemodynamic phenomenon is observed in spontaneous complete left branch block.
Actually, the classical pacing is deleterious for the myocardium and it is even used in hypertrophic obstructive cardiomyopathy, situation in which the functional impairment reflects on the desired reduction of the intraventricular gradient.
Problems of RVApical Pacing
Ventricular endocardial pacing in the RV apex (apical) is broadly used worldwide since the Sixties' and is the most widespread method of artificial cardiac pacing. However, although it has been revolutionary, safe and efficient to correct bradyarrhythmias, it induces a functional left branch block and promotes a serious desynchrony of the LV walls [15]. This undesirable "side effect" may cause or even worsen the HF, whose symptoms may not be completely taken into account since they are blunted by the increase in the cardiac output obtained by the bradyarrhythmia correction. However, if sustained , this condition promotes an undesirable myocardial remodeling which predisposes the patient to the appearance or worsening the HF, reduces the quality of life and leads to an increase in mortality, even in normal functioning AV sequential pacemakers [16].
In addition, several multi-center randomized trials have shown valuable information of the RV apical pacing damage, although they have been intended for another objective. MOST [17], DAVID [18] and MADIT-II [18] trials showed that the longer the time of RV apical pacing, the higher the HF, hospitalization, atrial fibrillation (AF), ventricular arrhythmias and mortality incidence.
These considerations make us to conclude that regardless these problems the RV endocardial pacing is the most frequently used treatment for bradyarrhythmias, although it causes several and unwanted consequences such as:
• Structural and histological remodeling of the myocardium
• Favors the AF
• Promotes or increases mitral regurgitation
• Promotes myocardial dysfunction
• Favors the HF progression
• Reduces quality of life
• Increases mortality
Alternative Pacing of the RV with one lead
Several areas of the RV may be safely stimulated by using modern active fixation leads (Figure 2).
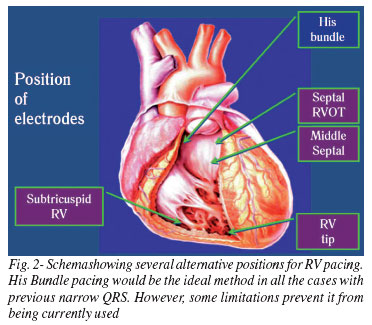
In an experimental trial comparing the hemodynamic result of different points of pacing in the RV [19], His Bundle pacing showed the best result as expected. However, the most important observation is that the worst outcomes were obtained from the RV apical pacing, namely conventional stimulation.
Although the stimulation of the His Bundle is more physiological and enables the maximum resynchronization with normal QRS which prevents the impairment of systolic and diastolic functions and mitral regurgitation, there are practical limitations which have been hindering its common use, such as: it is technically more difficult; it has higher thresholds; it needs special leads and introducers; it is susceptible to oversensing of atrial far-field and it may not be recommended in cases of previous His-Purkinje system lesions.
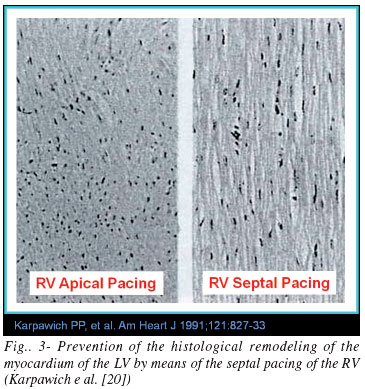
Considering the problems of apical pacing and to the limitations of currently using the His Bundle pacing it makes sense to pay greater attention to the septal pacing. lead, Modern active-fixation electrodes have low profile and are highly flexible which makes it relatively easy to lead the implantation by using the pre-shaped guide-wire to the midseptal or para-Hisian region. The electrocardiographic benefit is usually the first consequence observed during the pacemaker implant: the narrower QRS and the QRS axis closer to normal condition (positive or isodiphasic QRS in D1 and positive in D2, D3 and aVF). In addition, there is a significant echocardiographic improvement followed by clinical benefit that is usually reflected bythe reduction of the quality of life score.. These findings are strengthened by the experimental study of Karpawich et al. [20] who obtained great histological benefit from septal pacing in relation to apical pacing. They showed a very significant and udesirable histological remodeling by RV apical pacing that was prevented by the RV septal pacing (Figure 3).
Implantation Technique and Complications
Although it depends on a more accurate surgical procedure, no significant difficulty with the septal implantation or to para-Hisian implantation was observed since the technique and the radiological confirmation are taken into account during the lead placement. In general, the implantation was fast which did not increase the risk of infection and of radiological exposure.
One case of generator pocket hematoma caused by the chronic use of oral anticoagulants was observed. It was completely solved by surgical drainage before the hospital discharge. The sole complication directly associated with the septal implantation was an acute dislodgement which was corrected by lead repositioning during the same hospitalization. This fact deserves additional comments since they were associated with the use of an endocardial active fixation lead with a short screw. After this observation, this kind of lead was changed and only a model with longer fixation screw started to be used.
Threshold
The means of the unipolar and bipolar septal thresholds were 6.8% lower than the means of the unipolar and bipolar apical thresholds in the acute phase, but there was no statistically significant difference (P = 0.17 and 0.39 [twotailed paired t test] respectively) (Table 2). In the chronic phase (six months later), the means of the unipolar and bipolar septal thresholds were 44.4% (P =0.02) and 22.5% (P = 0.12) lower than the means of the unipolar and bipolar apical thresholds (Table2). In this case, although the means are quite similar, the chronic unipolar septal threshold was statistically lower (two-tailed paired t test). In relation to the pacing threshold, these results suggest that the septal pacing can be used or even preferred with no additional risk.
RWave
Septal and apical R waves of 21 patients with bifocal implantation in RV were compared. Four patients were completely dependent on the artificial pacing, which made it impossible to measure R wave. The results are shown in Table 2. It was observed that the unipolar and bipolar septal R waves are on average 1.1% and 0.8% higher and lower than the unipolar and bipolar apical R waves, respectively. However, these differences were not statistically significant, P = 0.80 and 0.93 (two-tailed paired t test).
These findings suggest that despite the septal position shows a slightly lower R wave, the means obtained with this cohort demonstrated excellent values: unipolar and bipolar 10.0 and 12.3 mV, respectively.
In the chronic phase, the means of the unipolar and bipolar septal R waves were 20% and 7.1% higher than the means of the unipolar and bipolar apical R waves, respectively, not showing statistically significant difference P = 0.09 and 0.50 (two-tailed paired t test) (Table 2). Accordingly, the amplitude of the R wave does not impose a limitation on septal implantation.
Impedances
Impedances were compared among 25 patients and it was observed that unipolar and bipolar septal impedances were on average 13.2% and 2.8% lower than the unipolar and bipolar apical impedances, respectively. However, these differences were not statistically significant, P = 0.09 and 0.34 (two-tailed paired t test). In the chronic phase, the means of the unipolar and bipolar septal impedances were respectively 0.6% and 5.1% lower and higher than the mean of unipolar and bipolar apical impedances, not showing statistically significant difference P = 0.8 and 0.24 (twotailed paired t test) (Table 2). These results demonstrate that in relation to the impedance, the septal pacing and the apical pacing are indifferent.
QRS Duration (QRSd)
Notwithstanding being a secondary endpoint of this study, an extremely relevant finding is that the QRS obtained with septal pacing was significantly narrower than the QRS resulted from the apical pacing (P < 0.01) (Table 3). Several studies demonstrate that in patients with cardiomyopathy, the longer the QRSd, the higher the mortality [21]. Furthermore, a number of evidences indicate that the myocardial desynchronization is proportional to the QRSd. In addition, recent observations corroborate less desynchrony of the LV with septal pacing [22].
Since these facts are now confirmed, we have definitely changed the ventricular pacing from conventional to septal pacing, starting from the 1990s when the advances in technology enabled the use of more suitable active-fixation leads [23].
In all the cases, the RV bifocal paced QRS was the narrowest, so that all the patients of the study could remain with this type of stimulation.
QRS AXIS
As secondary endpoint, electrical axis of paced QRS from septum and apex of the 25 bifocal implantations in the RV were also compared. The outcomes are shown in Table 4.
It was observed that the electrical axis of the septal paced QRS was positive and closer to the normal axis. The same situation was observed in relation to the QRS axis of bifocal pacing. Conversely, the mean axis of the QRS resulting from the apical pacing showed great difference in relation to the normal QRS axis. These differences were statistically significant, P = 0.0004 (two-tailed paired t test, measured between septal and apical positions); P1 = 0.0013 (two-tailed paired t test, measured between bifocal and apical positions); P2= 0.001(two-tailed paired t test, measured between bifocal and septal positions). These findings suggest that the electrical axis is more physiological when the QRS results from the septal in relation to the apical pacing. Hence, considering the electrical axis of the paced QRS, the septal pacing should be preferred in relation to the classical pacing, since the physiological progression of the myocardial activation reduces the histological remodeling of the wall of the LV [20], besides improving its hemodynamic performance [24].
CONCLUSION
The data yielded for this study demonstrate that the septal thresholds were slightly lower than the apical ones in an intrapatient comparison. This difference was not statistically significant. The electrical axis obtained in septal position was closer to the normal axis in relation to the apical position with important statistical significance.
The measurements of R waves and impedances in septal and apical positions did not show statistically significant differences. The QRS of the septal pacing was significantly narrower than the one produced by apical pacing.
These data show that the septal pacing may be regularly used with no electrophysiological objection.
Thus, according to the electrophysiological parameters studied here, besides there having no restrictions, there are even advantages in septal pacing, and it must be considered whenever as possible in cases with sole RV lead implantation.
REFERENCES
1. Xiao HB, Lee CH, Gibson DG. Effect of left bundle branch block on diastolic function in dilated cardiomyopathy. Br Heart J. 1991;66(6):443-7. [MedLine]
2. Auricchio A, Salo RW. Acute hemodynamic improvement by pacing in patients with severe congestive heart failure. Pacing Clin Electrophysiol. 1997;20(2 Pt 1):313-24. [MedLine]
3. Auricchio A, Stellbrink C, Block M, Sack S, Vogt J, Bakker P, et al. Effect of pacing chamber and atrioventricular delay on acute systolic function of paced patients with congestive heart failure. The Pacing Therapies for Congestive Heart Failure Study Group. The Guidant Congestive Heart Failure Research Group. Circulation. 1999;99(23):2993-3001. [MedLine]
4. Pignalberi C, Ricci RP, Santini M. Deleterious effects of apical right ventricular stimulation. Should we change our standard method of pacemaker implantation? Ital Heart J Suppl. 2005;6(10):635-48. [MedLine]
5. Hochleitner M, Hörtnagl H, Ng CK, Hörtnagl H, Gschnitzer F, Zechmann W. Usefulness of physiologic dual-chamber pacing in drug-resistant idiopathic dilated cardiomyopathy. Am J Cardiol. 1990;66(2):198-202. [MedLine]
6. Lamas GA, Orav EJ, Stambler BS, Ellenbogen KA, Sgarbossa EB, Huang SK, et al. Quality of life and clinical outcomes in elderly patients treated with ventricular pacing as compared with dual-chamber pacing. Pacemaker Selection in the Elderly Investigators. N Engl J Med. 1998;338(16):1097-104. [MedLine]
7. Pachón MJC, Pachón JC, Pachón MEI, Albornoz RN. Ventricular pacemaker syndrome. Europace. 2001;2(Supl B):B136.
8. Grines CL, Bashore TM, Boudoulas H, Olson S, Shafer P, Wooley CF. Functional abnormalities in isolated left bundle ranch block. The Effect of Interventricular Asynchrony. Am J Cardiol. 2000;86(12):1293-8. [MedLine]
9. Kormann DS, Jatene AD. Triângulo eletrodo vertebrodiafragmático no posicionamento de eletrodo endocavitário para marca-passo cardíaco. Arq Bras Cardiol. 1977;39(Supl. II):380.
10. Bakker PJ, Meijburg H, De Jonge N, Van Mechelen R, Wittkampf FH, Mower M. Beneficial effects of biventricular pacing in congestive heart failure. PACE. 1994;17(II):820.
11. Cazeau S, Leclercq C, Lavergne T, Walker S, Varma C, Linde C, et al; Multisite Stimulation in Cardiomyopathies (MUSTIC) Study Investigators. Effects of multisite biventricular pacing in patients with heart failure and intraventricular conduction delay. N Engl J Med. 2001;344(12):873-80. [MedLine]
12. Pachón JC, Pachón EI, Albornoz RN, Pachón JC, Kormann DS, Gimenes VM, et al. Ventricular endocardial right bifocal stimulation in the treatment of severe dilated cardiomyopathy heart failure with wide QRS. Pacing Clin Electrophysiol. 2001;24(9 Pt 1):1369-76. [MedLine]
13. Pachón Mateos JC, Pachón Mateos EI, Pachón Mateos JC. Right ventricular apical pacing: the unwanted model of cardiac stimulation? Expert Rev Cardiovasc Ther. 2009;7(7):789-99. [MedLine]
14. Gregoratos G, Abrams J, Epstein AE, Freedman RA, Hayes DL, Hlatky MA, et al American College of Cardiology/ American Heart Association Task Force on Practice Guidelines American College of Cardiology/American Heart Association/ North American Society for Pacing and Electrophysiology Committee. ACC/AHA/NASPE 2002 Guideline Update for Implantation of Cardiac Pacemakers and Antiarrhythmia Devices: Summary Article. A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/NASPE Committee to Update the 1998 Pacemaker Guidelines). J Cardiovasc Electrophysiol. 2002;13(11):1183-99. [MedLine]
15. Barold SS. Adverse effects of ventricular desynchronization induced by long-term right ventricular pacing. J Am Coll Cardiol. 2003;42(4):624-6. [MedLine]
16. Wilkoff BL, Cook JR, Epstein AE, Greene HL, Hallstrom AP, Hsia H, et al. Dual-chamber pacing or ventricular backup pacing in patients with an implantable defibrillator: the dual chamber and VVI implantable defibrillator (DAVID) trial. Dual chamber and VVI implantable defibrillator trial investigators. JAMA. 2000;288(24):3115-23.
17. Sweeney MO, Hellkamp AS, Ellenbogen KA, Greenspon AJ, Freedman RA, Lee KL, et al. Adverse effect of ventricular pacing on heart failure and atrial fibrillation among patients with normal baseline QRS duration in a clinical trial of pacemaker therapy for sinus node dysfunction. Circulation. 2003;107(23):2932-7. [MedLine]
18. Moss AJ, Zareba W, Hall WJ, Klein H, Wilber DJ, Cannom DS, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346(12):877-83. [MedLine]
19. Deshmukh PM, Romanyshyn M. Direct his-bundle pacing: present and future. Pacing Clin Electrophysiol. 2004;27(6 pt 2):862-70. [MedLine]
20. Karpawich PP, Justice CD, Chang CH, Gause CY, Kuhns LR. Septal ventricular pacing in the immature canine heart: a new perspective. Am Heart J. 1991;121(3 Pt 1):827-33. [MedLine]
21. Aro AL, Anttonen O, Tikkanen JT, Junttila MJ, Kerola T, Rissanen HA, et al. Intraventricular conduction delay in a standard 12-lead electrocardiogram as a predictor of mortality in the general population. Circ Arrhythm Electrophysiol. 2011;4(5):704-10. [MedLine]
22. Wang F, Shi H, Sun Y, Wang J, Yan Q, Jin W, et al. Right ventricular outflow pacing induces less regional wall motion abnormalities in the left ventricle compared with apical pacing. Europace. 2012;14(3):351-7. [MedLine]
23. Mateos JCP, Albornoz RN, Mateos EIP, Gimenez VM, Mateos JCP, Santos Filho ER, et al. Estimulação ventricular direita bifocal no tratamento da miocardiopatia dilatada com insuficiência cardíaca. Arq Bras Cardiol. 1999;73(6):485-91.
24. Karpawich PP, Mital S. Comparative left ventricular function following atrial, septal, and apical single chamber heart pacing in the young. Pacing Clin Electrophysiol. 1997;20(8 Pt 1):1983-8. [MedLine]
Article receive on Thursday, June 7, 2012
 All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license
All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license