Kamil Jurand PolokI; Jacek GórkaI; Jakub FronczekI; Karolina GórkaII; Bogusz KaczmarekI; Teresa IwaniecII; Paweł IwaszczukII; Jacek MusiałII; Wojciech SzczeklikII
DOI: 10.21470/1678-9741-2018-0238
ABSTRACT
Objective: The main goal of our study was to assess the impact of vascular procedures on the activity of hemostatic and fibrinolytic pathways.AAA = Abdominal aortic aneurysm
Ag = Antigen
APTEM, EXTEM, FIBTEM, INTEM, and HEPTEM = Types of ROTEM tests
BMI = Body mass index
CAT = Calibrated automated thrombography
CD40L = Cluster of differentiation 40 ligand
CoR = Ristocetin cofactor
ELISA = Enzyme-linked immunosorbent assay
FVIII = Factor VIII
IQR = Interquartile range
MI = Myocardial infarction
MINS = Myocardial injury after noncardiac surgery
PAD = Peripheral artery disease
PAP = Plasmin-antiplasmin complexes
ROTEM = Rotational thromboelastometry
sCD40L = Soluble cluster of differentiation 40 ligand
SD = Standard deviation
TIA = Transient ischemic attack
tPA = Tissue plasminogen activator
USA = United States of America
VTE = Venous thromboembolism
vWF = von Willebrand factor
INTRODUCTION
Every year more than 200 million noncardiac surgeries are performed worldwide. Despite developments in perioperative care, 1-2% of patients die within 30 days after the procedure. Cardiovascular complications including myocardial injury after noncardiac surgery (MINS), myocardial infarction (MI), stroke, and venous thromboembolism (VTE) are the most common perioperative events influencing mortality[1,2].
MINS is defined as a prognostically relevant troponin elevation within 30 days after surgery, which may present with or without electrocardiogram changes, ischemic symptoms, or cardiac contractility changes. MINS is estimated to occur in approximately 18% of patients and it is associated with the highest population-attributable risk of mortality among all perioperative complications[3-5]. MI, which is diagnosed in 3.1-3.9% of patients undergoing noncardiac surgeries, is a separate clinical entity, and its incidence among patients undergoing high-risk vascular surgeries shows no temporal changes and is invariably related to poorer prognosis[4,6-8]. Another complication associated with noncardiac surgery is the perioperative stroke, that occurs in 0.05-4% of patients in one-year observation, depending on the type of procedure[9,10]. Finally, VTE is a potentially life-threatening complication associated with vascular surgery, which occurs in approximately 1% of patients[11].
Patients undergoing vascular surgery, who commonly present with pronounced atherosclerosis, seem to be at a particularly high risk of both venous and arterial complications due to coexistence of multiple comorbidities and cardiovascular risk factors[12-14]. It was recently reported that alterations in preoperative levels of coagulation and fibrinolysis markers in serum are associated with a higher risk of developing MINS in patients undergoing vascular procedures[15]. Moreover, in a study by Górka et al.[16], it was shown that arterial complications are more common than venous complications in the perioperative period and are associated with higher mortality. We hypothesized that a major vascular surgery further potentiates a prothrombotic and hypofibrinolytic state as a consequence of intraoperative hypotension, hypoxemia, bleeding, injury, and pain[17,18]. Such changes could render patients more susceptible to cardiovascular complications.
There is a limited number of studies concerning the influence of vascular surgery on hemostasis. They were performed on small populations and are heterogeneous in terms of markers used as well as the number and timing of measurements. Moreover, their results are ambiguous and do not produce credible conclusions. Therefore, in this pilot study, we aimed to assess the impact of vascular surgery on hemostatic (factor VIII [FVIII], von Willebrand factor [vWF] activity, antithrombin), fibrinolytic (D-dimer, plasmin-antiplasmin complexes [PAP], tissue plasminogen activator [tPA]), and inflammatory (soluble cluster of differentiation 40 ligand [sCD40L]) markers’ levels. Additionally, we assessed whether the profile of changes in the aforementioned systems differs between patients undergoing procedures for abdominal aortic aneurysm (AAA) and those treated surgically for peripheral artery disease (PAD).
METHODS
We enrolled 38 consecutive patients undergoing open vascular surgery due to PAD or AAA in the Vascular Surgery Department of the St. John Grande Hospital, Krakow, Poland. Inclusion criteria in our study were: age ≥ 45 years, general or regional anesthesia for vascular surgery, and at least one-night hospital stay after the procedure. Exclusion criteria in this study were: carotid artery surgery, acute bypass graft thrombosis, cancer, renal failure defined as estimated glomerular filtration rate < 30 ml/min/1,73 m2, VTE within three months before surgery, and acute infection. Patients using heparin, oral anticoagulants, or antiplatelet drugs were included in the study.
Hemostatic, fibrinolytic, and inflammatory markers were measured before and 6-12 hours after the procedure using the following methods:
1) D-dimer - turbidimetric method (Innovance D-Dimer, Siemens, Marburg,
Germany);
2) tPA - enzyme-linked immunosorbent assay (ELISA) method (Zymutest t-PA
Antigen, Hyphen BioMed, Andresy, France);
3) Antithrombin activity - chromogenic method using BCS analyzer
(Innovance Antithrombin, Siemens, Marburg, Germany);
4) sCD40L - ELISA method (CD40 Quantikine ELISA Kit, R&D, Abingdon,
United Kingdom);
5) PAP complexes - ELISA method (Technozym PAP Complex, Technoclone,
Vienna, Austria);
6) vWF activity - turbidimetric method (BC von Willebrand Reagent,
Siemens, Marburg, Germany);
7) Activity of FVIII - one-stage clotting assay (Siemens, Marburg,
Germany).
All markers were measured in platelet-poor plasma.
The study was approved by the local ethics committee (Jagiellonian University Ethics Committee; approval number KBET/74/B/2010). The protocol complied with the Helsinki Declaration. All subjects studied signed written informed consent to participate.
Categorical variables were presented as numbers (percentages), whereas continuous variables were reported as medians (25-75 interquartile range), if not specified otherwise.
Preoperative and postoperative markers’ levels were compared using Wilcoxon signed-rank test. A P value of less than 0.05 was considered statistically significant.
We performed a comparison of pre- and postoperative levels of coagulation and fibrinolysis markers in the entire cohort as well as separately in groups depending on the type of surgery. Additionally, we compared pre- and postoperative antithrombin activity between patients who received and those who did not receive heparin within seven days prior to surgery.
A statistical analysis was performed with the use of Statistica 12 software (StatStoft®, Tulsa, Oklahoma, United States of America [USA]). Graphs were created in GraphPad Prism 7 software (GraphPad Software®, San Diego, California, USA).
RESULTS
We enrolled 38 patients with a mean age of 66.7 (standard deviation [SD]=7.6) years. There were 31 (81.6%) males in the study group. Reasons for surgery were AAA in 16 patients (42.1%) and PAD in 22 patients (57.9%). Detailed clinical and demographic characteristics of the study group can be found in Table 1. In the analysis of the entire cohort we found significant differences between preoperative and postoperative levels of all markers except for vWF:ristocetin cofactor (CoR) and PAP. We observed a significant increase in D-dimer and tPA levels and a decrease in FVIII, antithrombin, and cluster of differentiation 40 ligand (CD40L) levels. Postoperative antithrombin activity below the reference range of 80% was observed in 26 patients (68.4%). Detailed results can be found in Tables 2-4 and Figure 1.
| Characteristics | Study population (N = 38) | |
|---|---|---|
| Demographics | ||
| Sex; N (%): | Males | 31 (81.6%) |
| Females | 7 (18.4%) | |
| Age; mean (SD) | 66.7 (7.6) | |
| Reason for surgery | Abdominal aortic aneurysm; N (%) | 16 (42.1%) |
| Peripheral artery disease; N (%) | 22 (57.9%) | |
| Clinical characteristics | Overweight; N (%) | 20 (52.7%) |
| BMI*; mean (SD) | 25.5 (4.3) | |
| History of smoking; N (%) | 33 (86.8%) | |
| Packyears; median (quartiles) | 40.5 (23.5-56) | |
| Atrial fibrillation; N (%) | 4 (10.5%) | |
| Coronary artery disease; N (%) | 15 (39.5%) | |
| History of stroke/TIA † ; N (%) | 3 (7.9%) | |
| Heart failure; N (%) | 5 (13.2%) | |
| Chronic obstructive pulmonary disease; N (%) | 7 (18.4%) | |
| Hypertension; N (%) | 31 (81.6%) | |
| Diabetes mellitus; N (%) | 6 (15.8%) | |
| Oral anticoagulants; N (%) | 1 (2.6%) | |
| Heparin < 7 days prior to surgery; N (%) | 10 (26.3%) | |
| Antiplatelet drugs < 7 days prior to surgery; N (%) | 33 (86.8%) | |
| Marker | Marker level (median and quartiles) | P value | |
|---|---|---|---|
| Preoperative | Postoperative | ||
| Factor VIII (%) | 158.0 (139.4 – 188.8) | 103.3 (70.1 – 148.6) | <0.001 |
| vWF:CoR (%) | 140.6 (121.4 – 158.1) | 162.8 (111.4 – 219.8) | 0.17 |
| Antithrombin (%) | 92.1 (86.9 – 100.1) | 74.8 (69.3 – 84.9) | <0.001 |
| D-dimer (ng/ml) | 938.0 (697.0 – 3004.0) | 2406.0 (1218.0 – 5237.0) | 0.005 |
| PAP* (ng/ml) | 749.6 (626.3 – 1089.8) | 863.7 (621.7 – 1098.9) | 0.21 |
| tPA † (ng/ml) | 10.1 (8.7 – 14.1) | 12.8 (10.3 – 16.4) | 0.002 |
| sCD40L ‡ (pg/ml) | 9092.9 (7625.8 – 11363.6) | 1249.6 (694.4 – 2827.1) | <0.001 |
| Marker | Marker level (median and quartiles) | P value | |
|---|---|---|---|
| Preoperative | Postoperative | ||
| Factor VIII [%] | 151.8 (136.3 – 172.2) | 132.6 (85.1 – 150.2) | 0.04 |
| vWF:CoR [%] | 133.2 (112.9 – 150.6) | 200.0 (141.7 – 226.5) | 0.001 |
| Antithrombin [%] | 92.1 (89.3 – 97.7) | 75.3 (71.7 – 80.7) | <0.001 |
| D-dimer [ng/ml] | 3004.0 (938.0 – 4800.0) | 4164.0 (3102.5 – 6236.0) | 0.14 |
| PAP* [ng/ml] | 745.0 (608.0 – 1156.0) | 964.2 (797.5 – 1109.2) | 0.31 |
| tPA † [ng/ml] | 10.0 (9.3 – 10.9) | 11.7 (9.7 – 14.3) | 0.02 |
| sCD40L ‡ [pg/ml] | 8574.8 (7625.8 – 9182.1) | 1149.3 (676.7 – 1796.9) | 0.001 |
| Marker | Marker level (median and quartiles) | P value | |
|---|---|---|---|
| Preoperative | Postoperative | ||
| Factor VIII [%] | 174.1 (151.9 – 201.8) | 85.2 (48.7 – 148.6) | <0.001 |
| vWF:CoR [%] | 143.1 (135.2 – 167.3) | 138.0 (102.1 – 177.7) | 0.13 |
| Antithrombin [%] | 90.3 (82.8 – 101.4) | 71.4 (68.3 – 85.1) | <0.001 |
| D-dimer [ng/ml] | 768.5 (678.0 – 1302.0) | 1539.5 (704.0 – 2902.0) | 0.02 |
| PAP* [ng/ml] | 754.1 (669.7 – 1089.8) | 742.7 (578.3 – 1098.9) | 0.81 |
| tPA † [ng/ml] | 10.5 (6.7 – 17.7) | 14.9 (12.1 – 18.4) | 0.047 |
| sCD40L ‡ [pg/ml] | 9549.7 (7715.5 – 11918.0) | 1943.5 (771.6 – 3442.6) | <0.001 |
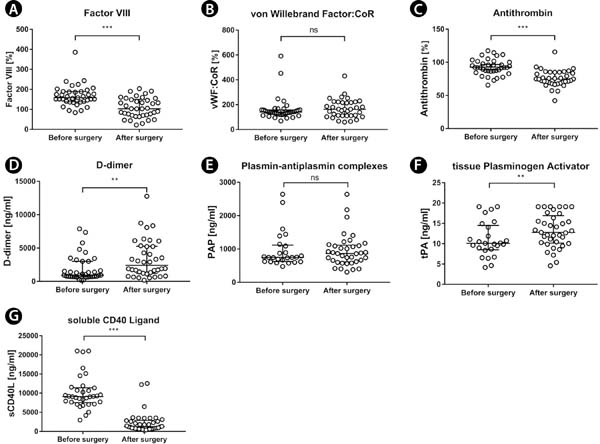
Additionally, we performed analyses in groups of patients undergoing surgeries for AAA and PAD. In the AAA group, we observed increase in vWF:CoR and tPA:antigen (Ag) levels, as well as a decrease in FVIII, antithrombin, and CD40L levels. There was no statistically significant difference between pre- and postoperative levels of D-dimer and PAP. In the PAD group, there was an increase in D-dimer and tPA levels and a decrease in FVIII, antithrombin, and CD40L levels. No perioperative differences in vWF and PAP levels were found.
There were no significant differences in the activity of antithrombin both preoperatively (89.8 vs. 92.1, P=0.52) and after surgery (71.1 vs. 75.8, P=0.34) between patients who received and those who did not receive heparin seven days prior to the procedure (Table 5).
| Patients receiving heparin (N=10) | Patients not receiving heparin (N=28) | P value | |
|---|---|---|---|
| Preoperative antithrombin activity; median (IQR) [%] | 89.8 (83.8 – 98.8) | 92.1 (87.4 – 99.6) | 0.52 |
| Postoperative antithrombin activity; median (IQR) [%] | 71.1 (66.4 – 74.5) | 75.8 (70.9 – 85.0) | 0.34 |
DISCUSSION
Our pilot study shows that vascular procedures have a significant impact on hemostatic and fibrinolytic systems. We documented important changes between pre- and postoperative levels of hemostatic (FVIII, vWF:CoR, antithrombin), fibrinolytic (D-Dimer, tPA:Ag), and inflammatory (CD40L) markers. Moreover, we observed several differences in the pattern of perioperative alterations in the aforementioned markers’ levels between patients undergoing procedures for PAD and those treated for AAA.
Changes in coagulation markers observed in the entire cohort do not justify drawing definite conclusions considering the impact of vascular procedures on hemostasis. Firstly, we observed a decrease in FVIII level that is possibly secondary to an increase in its consumption during augmented postoperative hemostasis. Interestingly, some previous reports suggest that FVIII levels tend to increase sharply in the following days, thereby contributing to postoperative hypercoagulable state[19]. Moreover, we documented an increase in vWF levels which was not statistically significant and is most probably related to its release from endothelium in response to vascular injury[20,21]. Importantly, a rise of vWF level above the reference range may be associated with a higher risk of thrombotic complications[22].
Finally, we observed a significant decrease in the antithrombin activity which is probably a consequence of an intraoperative heparin administration. However, this decrease resulted in a postoperative antithrombin deficiency in more than 60% of the study population. This condition can expose patients to a higher risk of thrombotic events due to temporary depletion of this natural anticoagulant. It is worth mentioning that a study by Ranucci et al.[23] showed that a decrease in antithrombin levels after cardiac surgery procedures with lower postoperative levels of antithrombin is associated with poorer patient outcomes. Interestingly, we did not observe any significant differences in pre- and postoperative antithrombin activity between patients who received and those who did not receive heparin within seven days before surgery.
Analysis of changes in the fibrinolytic system also presents a very complicated picture. We demonstrated a perioperative increase in D-dimer and tPA:Ag levels, but without any change between pre- and postoperative levels of PAP, which is a marker of overall plasmin production. D-dimer is a biomarker of ongoing clot formation and fibrinolysis and an increase in its level is a sign of higher fibrinolytic activity. On the other hand, elevation of tPA:Ag levels is an indication of hypofibrinolytic state. Interestingly, according to some larger previous studies, higher levels of both tPA:Ag and D-dimer are associated with an increased risk of cardiovascular events[24].
Analysis of subgroups offered some further valuable insights. Firstly, there was an increase in vWF:CoR level in the AAA group that was not observed in PAD group, which might suggest more pronounced activation of coagulation system associated with open aortic repair compared to surgeries on peripheral arteries. Moreover, D-dimer levels increased perioperatively in patients undergoing surgery for PAD, while in the other group there was no statistically significant change in its level, which may represent stronger triggering of fibrinolysis associated with these procedures. However, an assessment of absolute values offers an important observation - D-dimer levels are four times higher preoperatively and more than two times higher postoperatively in patients in the AAA group than in the PAD group. This may indicate that hyperfibrinolysis is more pronounced in the AAA group, while it is the PAD surgery that significantly activates this pathway.
sCD40L is considered a proinflammatory and prothrombotic molecule. Its contribution to the development of atherosclerosis is strongly suggested and increased levels of sCD40L are associated with higher cardiovascular risk[25-28]. In current study, we observed a decrease in sCD40L level after both types of vascular procedures, which corroborates the study by Yip et al. showing similar change in sCD40L in patients undergoing coronary stenting. The authors hypothesized that it may either be a result of recovery from acute inflammation or be associated with reduced progression of inflammation in the plaque lesion[29]. This explanation can probably also be applied to our study. However, for the time being, clinical significance of this decrease in sCD40L level remains unclear.
There is a paucity of studies comparing preoperative and postoperative levels of hemostatic and fibrinolytic activation markers in patients undergoing vascular surgeries. Their results are difficult to compare due to heterogeneous methodologies, i.e., different markers and blood collection times. Nevertheless, these few published papers that we found suggest that surgical procedures change the activity of both hemostatic and fibrinolytic systems, although the direction of changes varies between the studies, especially among fibrinolytic markers[30-32].
We decided to perform this pilot to obtain preliminary results and evaluate our methodology in order to optimally design the main study. First, credible assessment of changes in hemostatic and fibrinolytic activity and its association with potential postoperative complications requires larger and more homogenous study population. Moreover, in our opinion, the present and previous results suggest that the use of serum markers alone may be insufficient to reliably assess perioperative changes in hemostatic and fibrinolytic systems. Thus, the use of more up-to-date, dynamic methods, such as rotational thromboelastometry (ROTEM) or calibrated automated thrombography (CAT), should warrant more plausible results. ROTEM is an established method of real-time assessment of whole blood hemostasis. It encompasses several tests that give a unique opportunity to separately assess extrinsic pathway (EXTEM), intrinsic pathway (INTEM), to inhibit platelet function and therefore assess effectiveness of fibrin polymerization (FIBTEM), to exclude hyperfibrinolysis (APTEM), and to eliminate influence of heparin on blood coagulation (HEPTEM)[33].
CAT is another dynamic method useful in the evaluation of hemostatic and fibrinolytic systems’ activity, based on real-time recording of thrombin generation[34]. Both tools would be particularly useful in patients undergoing vascular surgery who routinely receive heparin, anticoagulants, and blood products transfusions, which makes the assessment of hemostasis even more complex.
Limitation
Our study has several important limitations. First, the study group is relatively small and heterogeneous. Also, an association between perioperative changes in hemostatic and fibrinolytic markers and observed complications could not be thoroughly investigated due to a low number of events. Moreover, most of our patients experienced postoperative bleeding and received an antiplatelet drug or heparin prior to surgery which may have influenced some of the studied markers’ levels. Finally, employing intraoperative and additional postoperative measurements would markedly improve our understanding of perioperative alterations in hemostatic and fibrinolytic systems’ activities.
CONCLUSION
We demonstrated that major vascular surgeries lead to changes in both hemostatic and fibrinolytic activities, however their direction remains unclear and differs depending on the type of performed surgery. Observed changes in studied markers’ levels may contribute to an increased risk of MINS, MI, stroke, and VTE in the perioperative period. Further studies performed on larger populations and utilizing dynamic methods are warranted.
REFERENCES
1. Weiser TG, Regenbogen SE, Thompson KD, Haynes AB, Lipsitz SR, BerryWR, et al. An estimation of the global volume of surgery: a modelling strategybased on available data. Lancet. 2008;372(9633):139-44.doi:10.1016/S0140-6736(08)60878-8.
2. Kristensen SD, Knuuti J. New ESC/ESA guidelines on non-cardiacsurgery: cardiovascular assessment and management. Eur Heart J.2014;35(35):2344-5. doi:10.1093/eurheartj/ehu285.
3. Botto F, Alonso-Coello P, Chan M, Villar J, Xavier D, Srinathan S,et al. Myocardial injury after noncardiac surgery: a large, international,prospective cohort study establishing diagnostic criteria, characteristics,predictors, and 30-day outcomes. Anesthesiology. 2014;120(3):564-78.doi:10.1097/ALN.0000000000000113.
4. Devereaux PJ, Biccard BM, Sigamani A, Xavier D, Chan MT V.,Srinathan SK, et al. Association of postoperative high-sensitivity troponinlevels with myocardial injury and 30-Day mortality among patients undergoingnoncardiac surgery. JAMA. 2017;317(16):1642-51.doi:10.1001/jama.2017.4360. [MedLine]
5. Biccard BM, Scott DJA, Chan MT V, Archbold A, Wang C-Y, Sigamani A,et al. Myocardial injury after noncardiac surgery (MINS) in vascular surgicalpatients: a prospective observational cohort study. Ann Surg.2018;268(2):357-63. doi:10.1097/SLA.0000000000002290. [MedLine]
6. Devereaux PJ, Goldman L, Cook DJ, Gilbert K, Leslie K, Guyatt GH.Perioperative cardiac events in patients undergoing noncardiac surgery: a reviewof the magnitude of the problem, the pathophysiology of the events and methodsto estimate and communicate risk. CMAJ. 2005;173(6):627-34.doi:10.1503/cmaj.050011.
7. Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD,et al. Third universal definition of myocardial infarction. Circulation.2012;126(16):2020-35. doi:10.1161/CIR.0b013e31826e1058.
8. Juo Y-Y, Mantha A, Ebrahimi R, Ziaeian B, Benharash P. Incidence ofmyocardial infarction after high-risk vascular operations in adults. JAMA Surg.2017 15;152(11):e173360. doi:10.1001/jamasurg.2017.3360.
9. Axelrod DA, Stanley JC, Upchurch GR, Khuri S, Daley J, Henderson W,et al. Risk for stroke after elective noncarotid vascular surgery. J Vasc Surg.2004;39(1):67-72. doi:10.1016/j.jvs.2003.08.028. [MedLine]
10. Ng JL, Chan MT V, Gelb AW. Perioperative stroke in noncardiac,nonneurosurgical surgery. Anesthesiology. 2011;115(4):879-90.doi:10.1097/ALN.0b013e31822e9499.
11. Aziz F, Patel M, Ortenzi G, Reed AB. Incidence of postoperative deepvenous thrombosis is higher among cardiac and vascular surgery patients ascompared with general surgery patients. Ann Vasc Surg. 2015;29(4):661-9.doi:10.1016/j.avsg.2014.11.025.
12. Hirsch AT, Criqui MH, Treat-Jacobson D, Regensteiner J, Creager M,Olin J, et al. Peripheral arterial disease detection, awareness, and treatmentin primary care. JAMA. 2001;286(11):1317-24.doi:10.1001/jama.286.11.1317.
13. Kullo IJ, Rooke TW. Peripheral artery disease. N Engl J Med.2016;374(9):861-71. doi:10.1056/NEJMcp1507631.
14. Bani-Hani M, Titi M, Al-khaffaf H. Deep venous thrombosis afterarterial surgery: a literature review. Eur J Vasc Endovasc Surg.2008;36(5):565-73. doi:10.1016/j.ejvs.2008.07.007.
15. Górka J, Polok K, Iwaniec T, Górka K, Wludarczyk A, Fronczek J, etal. Altered preoperative coagulation and fibrinolysis are associated withmyocardial injury after non-cardiac surgery. Br J Anaesth. 2017;118(5):713-9.doi:10.1093/bja/aex081. [MedLine]
16. Górka J, Polok K, Fronczek J, Górka K, Kózka M, Iwaszczuk P, et al.Myocardial injury is more common than deep venous thrombosis after vascularsurgery and is associated with a high one year mortality risk. Eur J VascEndovasc Surg. 2018;56(2):264-70.doi:10.1016/j.ejvs.2018.02.005. [MedLine]
17. Siemens HJG, Brueckner S, Hagelberg S, Wagner T, Schmucker P. Courseof molecular hemostatic markers during and after different surgical procedures.J Clin Anesth. 1999;11(8):622-9.doi:10.1016/S0952-8180(99)00119-1.
18. Payen JF, Baruch M, Horvilleur E, Richard M, Gariod T, Polack B.Changes in specific markers of haemostasis during reduction mammoplasty. Br JAnaesth . 1998 ;80(4):464-6. Available from:
19. Kahlon A, Grabell J, Tuttle A, Engen D, Hopman W, Lillicrap D, etal. Quantification of perioperative changes in von Willebrand factor and factorVIII during elective orthopaedic surgery in normal individuals. Haemophilia.2013;19(5):758-64. doi:10.1111/hae.12185.
20. Gil-Bazo I, Gil Bazo I, Catalán Goni V, Catalán González V, AlonsoGutiérrez A, Rodríguez Rodríguez J, et al. Impact of surgery and chemotherapy onvon Willebrand factor and vascular endothelial growth factor levels incolorectal cancer. Clin Transl Oncol. 2005;7(4):150-5.doi:10.1007/BF02708752.
21. Vischer UM. von Willebrand factor, endothelial dysfunction, andcardiovascular disease. J Thromb Haemost. 2006;4(6):1186-93.doi:10.1111/j.1538-7836.2006.01949.x.
22. Spiel AO, Gilbert JC, Jilma B. Von Willebrand factor incardiovascular disease: focus on acute coronary syndromes. Circulation.2008;117(11):1449-59. doi:10.1161/CIRCULATIONAHA.107.722827.
23. Ranucci M, Frigiola A, Menicanti L, Ditta A, Boncilli A, Brozzi S.Postoperative antithrombin levels and outcome in cardiac operations. Crit CareMed. 2005;33(2):355-60. doi:10.1097/01.CCM.0000153409.55645.58.
24. Gorog DA. Prognostic value of plasma fibrinolysis activation markersin cardiovascular disease. J Am Coll Cardiol. 2010;55(24):2701-9.doi:10.1016/j.jacc.2009.11.095.
25. Lobbes MBI, Lutgens E, Heeneman S, Cleutjens KBJM, Kooi ME, vanEngelshoven JMA, et al. Is there more than C-reactive protein and fibrinogen?.The prognostic value of soluble CD40 ligand, interleukin-6 and oxidizedlow-density lipoprotein with respect to coronary and cerebral vascular disease.Atherosclerosis. 2006;187(1):18-25.doi:10.1016/j.atherosclerosis.2005.11.005. [MedLine]
26. Antoniades C, Bakogiannis C, Tousoulis D, Antonopoulos AS,Stefanadis C. The CD40/CD40 Ligand system: linking inflammation withatherothrombosis. J Am Coll Cardiol. 2009;54(8):669-77.doi:10.1016/j.jacc.2009.03.076.
27. Schonbeck U, Varo N, Libby P, Buring J, Ridker PM. Soluble CD40L andcardiovascular risk in women. Circulation. 2001;104(1524-4539):2266-8.doi:10.1161/hc4401.099447.
28. Heeschen C, Dimmeler S, Hamm CW, Brand MJ Van Den, Boersma E, ZeiherAM, et al. Soluble CD40 Ligand in acute coronary syndromes. N Engl J Med.2003;348(12):1104-11. doi:10.1056/NEJMoa022600.
29. Yip H-K, Wu C-J, Yang C-H, Chang H-W, Fang C-Y, Hung W-C, et al.Serial changes in circulating concentrations of soluble CD40 ligand andC-reactive protein in patients with unstable angina undergoing coronarystenting. Circ J . 2005 ;69(8):890-5. Availablefrom:
30. Davies RSM, Abdelhamid M, Wall ML, Vohra RK, Bradbury AW, Adam DJ.Coagulation, fibrinolysis, and platelet activation in patients undergoing openand endovascular repair of abdominal aortic aneurysm. J Vasc Surg.2011;54(3):865-78. doi:10.1016/j.jvs.2011.04.010.
31. Gibbs NM, Patrick G, Crawford M, Michalopoulos N. Postoperativechanges in coagulant and anticoagulant factors following abdominal aorticsurgery. J Cardiothorac Vasc Anesth. 1992;6(6):680-5.doi:10.1016/1053-0770(92)90051-8.
32. keda U, Shimada K. Activated state of blood coagulation andfibrinolysis in patients with abdominal aortic aneurysm. Am J Surg .1999 ;177(6):527. Available from:
33. Whiting D, Dinardo JA. TEG and ROTEM: Technology and clinicalapplications. Am J Hematol. 2014;89(2):228-32.doi:10.1002/ajh.23599.
34. Hemker HC, Giesen P, AlDieri R, Regnault V, De Smed E, Wagenvoord R,et al. The calibrated automated thrombogram (CAT): a universal routine test forhyper- and hypocoagulability. Pathophysiol Haemost Thromb. 2002;32(5-6):249-53.doi:10.1159/000073575.
Financial support: This work was supported by a grant from the Polish Ministry of Science and Higher Education. Grant number NN402083939 and DI2011023141.
No conflict of interest.
Authors' roles & responsibilities
KJP Conception and design; analysis and interpretation; data collection; writing the article; critical revision of the article; statistical analysis; final approval of the version to be published
JG Conception and design; analysis and interpretation; data collection; writing the article; critical revision of the article; statistical analysis; final approval of the version to be published
JF Conception and design; analysis and interpretation; data collection; writing the article; critical revision of the article; final approval of the version to be published
KG Conception and design; analysis and interpretation; data collection; writing the article; critical revision of the article; final approval of the version to be published
BK Conception and design; analysis and interpretation; data collection; writing the article; critical revision of the article; final approval of the version to be published
TI Conception and design; laboratory measurements; analysis and interpretation; writing the article; critical revision of the article; final approval of the version to be published
PI Conception and design; data collection; writing the article; critical revision of the article; final approval of the version to be published
JM Conception and design; writing the article; critical revision of the article; final approval of the version to be published
WS Conception and design; analysis and interpretation; data collection; writing the article; critical revision of the article; statistical analysis; final approval of the version to be published
Article receive on Monday, August 13, 2018
Article accepted on Tuesday, November 20, 2018
 All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license
All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license