Roberto T. Sant'AnnaI; James FracassoII; Felipe H. ValleI; Iran CastroIII; Nance B. NardiIV; João Ricardo M. Sant'AnnaIII; Ivo Abrahão NesrallaI; Renato A. K. KalilIII
DOI: 10.5935/1678-9741.20140091
BMMC: Bone marrow mononuclear cells
DCM: Dilated cardiomyopathy
LVEF: Left ventricular ejection fraction
MLHF: Minnesota Living with Heart Failure
MRI: Magnetic resonance cardiac imaging
NIDCM: Non-ischemic dilated cardiomyopathy
NYHA: New York Heart Association
PO: Post-operatively
INTRODUCTION
Dilated cardiomyopathy is a leading cause of heart failure and the primary indication for heart transplantation[1,2]. The prevalence of this disease tends to increase according to population ageing and survival improvement achieved through advances in pharmacological treatment and implantable devices[3]. However, current therapeutic approaches are palliative in the sense that they are unable to directly address the underlying problem of the loss of cardiac tissue[4].
Cell therapy with bone marrow mononuclear cells (BMMC) has been associated with beneficial effects in patients with acute myocardial infarction and ischemic heart failure. Data for non-ischemic dilated cardiomyopathy (NIDCM), however, is more limited[5-7]. Experimental studies suggest that stem cells improve heart function through both paracrine regulation of cytokines production and cell transdifferentiation, albeit in a small proportion of the transplanted cells[8,9]. Two small trials, using BMMC through intracoronary route, showed small, but significant increase in left ventricular ejection fraction (LVEF) after treatment[5,6]. Recently, intracoronary injection of CD34+ was associated with medium-term improvement of ventricular function, exercise tolerance, and long-term survival in randomized trial of patients with DCM[7].
Intracoronary injection is associated with a small percentage of transplanted cells effectively retained in the myocardium, which could limit results when that route is used for cell therapy[10]. In a pilot study, we found that direct application of BMMC through a small thoracotomy was safe and associated with a significant, although transitory, increase in LVEF in patients with non-ischemic dilated cardiomyopathy (NIDCM)[11]. In the present study, we tested the hypothesis that direct intramyocardial injection of BMMC in patients with NIDCM could improve LVEF assessed by cardiac magnetic resonance imaging (MRI) and echocardiogram, physical capacity evaluated by the six-minute walk test (6WT), and heart failure class functional (NYHA), when compared to conventional treatment.
METHODS
Study design
We conducted an open, parallel-group, explanatory randomized study in a single center to evaluate the safety and efficacy of BMMC in patients NIDCM. Thirty patients that fulfilled the criteria for inclusion were randomly assigned to each of the groups by means of a computer software for simple randomization: BMMC or control, in a 2:1 proportion.
After randomization, functional status of both groups was assessed at baseline using (6WT), New York Heart Association (NYHA) and Minnesota Living with Heart Failure (MLHF) questionnaires, which had been previously validated for the Brazilian population[12]. We used cardiac magnetic resonance imaging (MRI) to measure left ventricular diameters and ejection fraction. Functional status was reassessed every three months and MRI repeated three and nine months after inclusion. All patients were treated by the same physician at a dedicated outpatient clinic, according to ACC/AHA Guidelines for the Diagnosis and Management of Heart Failure in Adults[1]. Functional status was evaluated by a separate team. In this article, we present the results of the first year of follow-up. We were unable to perform a blind study because of the invasive nature of the procedure proposed. The study was submitted to the National Comission in Research (CONEP) and approved. All patients signed an informed consent.
Patients in the BMMC group, in addition to conventional treatment, were submitted to BMMC transplantation as described below up to one month after randomization. In this group, we also conducted flow citometry and imunohistochemistry to characterize bone marrow cells.
Our primary objective was to evaluate changes in left ventricular ejection fraction assessed with MRI. Secondary objectives consisted of evaluation of: 1) Safety; 2) New York Heart Association (NYHA) functional class and quality of life measured with Minnesota Living with Heart Failure Questionnaire; 3) Effects in mortality; 4) Left ventricular diameters using MRI.
Inclusion and exclusion criteria
The study was approved by the Ethics Review Board of Instituto de Cardiologia do Rio Grande do Sul/ Fundação Universitária de Cardiologia (IC/FUC), as well as by the National Ethics Committee in Research of Brazil, under protocol number 10376 and registered in ClinicalTrials.gov under number: NCT00743639. Both the study and the registry were conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from each patient.
We screened for patients with heart failure, LVEF less then 35% and functional class III or IV, despite full medical treatment. Specific inclusion criteria were: (1) Age between 20 and 65 years; (2) Diagnosis of non-ischemic cardiomyopathy for at least 12 months before enrollment; (3) Coronary angiography with normal coronary arteries, which defined cardiomyopathy as non-ischemic for the purposes of this trial; (4) negative serology for Chagas Disease; and (5) Absence of other significant systemic disease limiting mid-term prognosis. We excluded patients with: (1) Documented episodes of ventricular tachycardia; (2) Moderate to severe mitral regurgitation or any other significant valve disease; (3) History of myocardial infarction; and (4) Previous cardiac surgery.
Bone marrow mononuclear cells isolation and transplantation
Approximately four hours before the operation and with the patient under sedation, a volume of about 80 mL of bone marrow was aspirated from the anterior iliac crest and placed in an anticoagulated medium. Mononuclear cell fraction was separated by density centrifugation over Ficoll-Hypaque-1077 (Sigma Diagnostics, St Louis, MO) and washed in a heparinized saline solution containing 5% autologous serum. Cells were counted and suspended in 5 mL saline solution for intramyocardial injection. A small fraction was utilized for sterility and viability tests and for immunophenotyping. Viability greater than 90% was considered acceptable.
For detection of surface antigens the cells were trypsinized, centrifuged, and incubated for 30 minutes at 4ºC with phycoerythrin (PE)- or fluorescein isothiocyanate (FITC)-conjugated antibodies against human CD3, CD4, CD14, CD34, CD38 and CD45 (Pharmingen BD, San Diego, CA). Excess antibody was removed by washing. The cells were analyzed using a FACScalibur cytometer equipped with 488 nm argon laser (Becton Dickinson, San Diego, CA) with the CellQuest software. At least 10,000 events were collected. WinMDI 2.8 software was used for building histograms.
Surgical approach was through a left mini-thoracotomy, consisting of an approximately 5 cm incision in the anterolateral portion of the fifth left intercostal space to expose the pericardium. A T-shaped pericardial incision was made to access the free wall of the left ventricle. Coronary arteries were identified and the cell suspension was directly injected, using a 21F Butterfly needle that was introduced about 5mm intramyocardially and connected to an extension managed by the surgical assistant. Twenty 0.25 mL injections were given in the myocardium and in the anterior, lateral, posterior, and apical faces of the left ventricular free wall. After reviewing the hemostasis, pericardium was approximated, the thoracic cavity was drained and the chest wall closed.
Echocardiogram
Two-dimensional echocardiograms were obtained for measuring end-systolic volume (ESV), end-diastolic volume (EDV), and LVEF, according to standard protocols. All exams were performed by the same person, who was unaware of the treatment group and was not part of the study team. It should be noted, however, that treated patients had a visible scar in the left chest, which made it impossible to guarantee blindness to the echo examinator.
Magnetic Resonance Imaging
All MRI image analyses were performed by the same investigator, which was blind to the assignment of the patient. We used a 1.5-T scanner (GE Excite HDx) with ECG gating and a 4-element phased-array surface coil. Scan planes were planned according to standard procedures. Endocardial and epicardial borders were traced in all end-diastolic and end-systolic short-axis slices to determine LV end-diastolic and end-systolic volumes (LVEDV and LVESV). Global LVEF was calculated [(LVEDV-LVESV)/LVEDV]/100 LVEDV. Systolic ventricular wall thickening was calculated for different heart segments and it was used for an already published sub-study[11].
Statistical analysis
Continuous variables are presented as mean±SD, if not stated otherwise. Categorical variables were compared with the x2 test or Fisher's exact test. Statistical comparisons between initial and follow-up data were performed in a nonparametric paired fashion using the Wilcoxon signed rank test. Nonparametric Mann-Whitney U and Kruskal-Wallis tests were used to compare continuous and categorical variable, as well as the results between different groups. All tests were performed as 2-sided tests at a significance level of 0.05.
In the first three months of follow-up, 25% of the patients from the BMMC group had either died or been withdrawn from the study, resulting in a decrease in the number of treated cases available for late follow-up. Since those patients had a lower mean ejection fraction than the whole group (18.26 % vs. 21.75%), we excluded them from comparative analysis, in order to avoid overestimation of treatment effect. In other words, outcome analysis was performed in as-treated basis, not as intention-to-treat.
RESULTS
Baseline
Baseline characteristics are detailed in Table 1. There were no significant differences in baseline data between BMMC and control groups. All patients had severe heart failure and were highly symptomatic by the time of inclusion, despite maximum medical therapy.

Cell analysis
A mean of 1.06±0.43 x 108 mononuclear cells per patient were available for injection using this method. Cell viability was greater than 99%, and fungal and bacterial cultures were negative.
Cells from the first five samples were analyzed in greater detail. Immunophenotyping of the mononuclear cell fraction showed the following composition: CD34+ cells (1.5±0.7%), CD45+ (74.6±8.5%), CD14+ (8.4±4.7%), CD3+CD4+ (22.8±4.6%), CD3+CD8+ (8.2±6.1%), and CD34+CD38- cells (0.7±0.5%).
Procedural safety and clinical outcomes
Surgical procedure was effectively performed in 19 patients, since one patient asked to be withdrawn soon after randomization. Table 2 summarizes surgical results. Four subjects died in the first month after the procedure. Two died of refractory cardiogenic shock in the first 72 hours post-operatively (PO). One of them presented with cardiac tamponade 7 hours after surgery and was submitted to reoperation. The patient's hemodynamic condition continued to deteriorate and he died due to cardiogenic shock 3 days PO. Another patient died on the 15th day PO due to incessant ventricular tachycardia. Finally, one patient died on the 28th day PO: he had been discharged from hospital, but was readmitted one week later due to heart failure, which was refractory to treatment. All of the patients above had LVEF below 21% before operation.
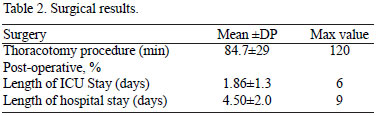
Three patients from the BMMC group died between 30 days and 12 months after surgery. In two patients, death was sudden, at 10 and 11 months PO. Another patient died 6 months PO due to heart failure.
In the control group, one patient died three months after randomization due to heart failure and another patient was lost for follow-up two months after randomization. There was no statistically significant difference in safety outcomes between BMMC and control groups: Fischer´s test showed no difference in mortality between groups in the first 30 days (P=0.371) or up to 12 months (P=1.000).
Effects of BMMC on LV Function
MRI evaluations
Patients of the BMMC group maintained LVEF at 3 months, from 27.80±6.86% to 30.13±9.06% (P=0.08), and at 9 months (28.78%, P=0.77). The control group also maintained systolic function during follow-up (28±4.32% vs. 27.4±7.4%, at 3 months, P=0.79, and vs. 29.57±4.50 at 9 months, P=0.46, respectively). Differences between groups were non-significant.
Both groups maintained end-systolic and end-diastolic volumes during follow-up. (Table 3).
Echocardiographic evaluations
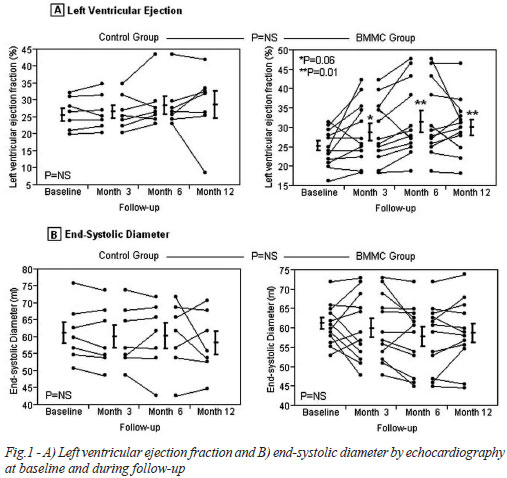
Patients of the BMMC group had a significant improvement in left ventricular ejection fraction 3 months after the procedure, from 25.09±3.98 to 30.94±9.16 (P=0.01). This benefit was maintained after one year of follow-up (30.07±7.25, P=0.001). The control group has showed no change in the same period (26.1±4.4 vs. 26.5±4.7 and 30.2±7.39, P=0.25 and 0.10, respectively). Differences between treated and control groups, however, were not statistically significant. Left ventricular fractional shortening evaluation showed similar results. End-systolic and end-diastolic diameters had a small reduction in both groups. (Figure 1, Table 4).
Quality of life and 6-minute walk test
Quality of life, evaluated by MLHFQ (Minnesota Living with Heart Failure Questionnaire), improved significantly in the BMMC group three months after the procedure (69.00±20.71 vs. 43.13±27.92 points, P=0.005), a benefit maintained during 6 and 12 months of follow-up (48.92±24.26 points, P=0.018; 37.08±21.15 points, P=0.001; respectively). Patients of the control group showed a non-significant improvement, with smaller differences between baseline and 12 months follow-up (minus 31.92 points for BMMC group and minus 7.16 points for Control group). Differences between groups were not statistically significant. Functional class, evaluated using NYHA Classification, showed similar results with a statistically significant improvement in the BMMC group, no change in the control group. However, once again, there were no statistically significant differences between groups. (Figure 2, Table 5).
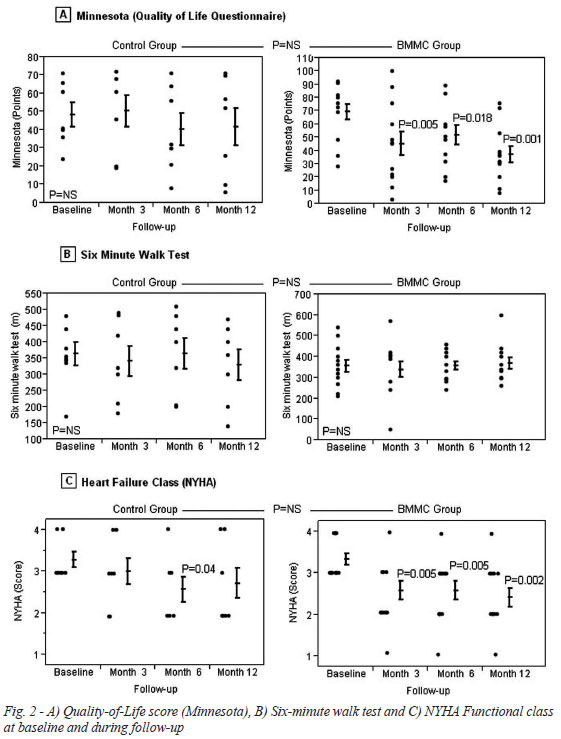
Six-minute-walk test results were maintained in the BMMC group (from 348.00±93.51min in baseline to 370.41±91.56 min after 12 months, P=0.66) and in control group (from 361.25±90.78 min in baseline to 330.00±123.42 min after 12 months, P=0.66). Again, differences between groups were not statistically significant. (Table 5).
DISCUSSION
In this study, we tested the hypothesis that transplantation of BMMC through a small thoracotomy could improve left ventricular function in patients with refractory heart failure due to NIDCM. Our main findings were: (1) Mortality associated with the procedure was directly related to preoperative disease severity. In particular, patients with LVEF below 21% had a poor surgical result. Early deaths were related to postoperative pump-failure and congestive heart failure, while deaths at medium term were sudden, probably caused by cardiac arrhythmias. These results differ from our previous pilot study, in which there had been no deaths[11]. (2) Survivors treated with BMMC maintained left ventricular function relative to baseline according to MRI. (3) Overall, patients had symptomatic improvement during follow-up, which was more pronounced in the BMMC group, but there was no statistical difference between groups in the outcomes studied.
The seven deaths (35%) observed in patients who received BMMC were due to pump-failure in the early post-operative period (2 cases; 28.5%), congestive heart failure (2 cases, 28.5%) and documented ventricular arrhythmia or sudden death (3 cases, 42.8%). Using the Seattle Heart Failure Model, a tool with excellent accuracy for predicting survival among patients with heart failure[13], we could anticipate one-year mortality between 20% and 25%. Incidence of death attributable to arrhythmia (42,8%) was higher than expected for patients in NYHA functional class III (10.5%) or IV (18.6%) at one year[14].
This higher than expected risk of ventricular tachyarrhythmia after surgery could be related to the intrinsic high risk of any procedure in patients with severe heart failure or to a more specific mechanism secondary to BMMC injection. Stem cell pro-arrhythmic potential is a matter of intense debate[15,16]. Clinically, bone marrow progenitor cells were not associated with increased risk of arrhythmias using different routes of delivery in different clinical scenarios[17-19]. Yet, experimental studies have shown that mesenchymal stem cells (MSCs) implantation altered cardiac conduction[20] and created areas of slow conduction, predisposing it to reentry episodes[21,22]. Intramyocardial injections can create clusters of cells leading to heterogeneity in conduction and have been associated with greater risk of ventricular premature complexes than intracoronary injection[23]. How this translates into clinical practice, where patients are treated with anti-arrhythmic drugs, needs to be further clarified. At this point, however, we cannot dismiss the possibility of a pro-arrhythmic effect related the newly implanted cells.
BMMC treated patients who survived for more than one month after the procedure showed significant improvements in both symptoms and quality of life. Left ventricular ejection fraction was maintained one year after the procedure (P=0.77). Results with echocardiography are more pronounced as they showed improvement in LVEF of patients treated with BMMC after 6 months (from 25.09±3.98 to 30.94±9.16, P=0.01). Nevertheless, since operators were not blind for patients' group, results from this method should be interpreted with caution.
Clinical experience with mononuclear cells in patients with DCM is limited. Fischer-Rasokat et al performed infusion of BMMC in 33 patients with DCM using an over-the-wire balloon catheter in the left descending coronary artery[8]. After 3 months, regional wall motion of the target area and global left ventricular ejection fraction had a small (about 10%) but statistically significant improvement. Increase of regional contractile function was directly related to the functionality of infused cells as measured by their colony-forming capacity. Safety data was remarkably good, with no procedure related complications and no deaths, stroke or myocardial infarction up to one year follow-up. Compared to our study, patients were less symptomatic (67% in NYHA class II) and had better ventricular function (mean LEVF=0.2%).
Vrtovec et al.[7] conducted a study with 110 patients with NIDCM, which were randomized to receive CD34+ stem cell transplantation and 55 received no cell therapy. Patients underwent myocardial scintigraphy and cells were injected in the artery supplying segments with the greatest perfusion defect. At 5 years, stem cell therapy was associated with increased LVEF (from 24.3±6.5% to 30.0±5.1%; P=0.02), increased 6-minute walk distance (from 344±90 m to 477± 30 m; P<0.001), and decreased N-terminal B-type natriuretic peptide (from 2322±1234 pg/mL to 1011±893 pg/mL; P<0.01). Left ventricular ejection fraction improvement was more significant in patients with higher myocardial homing of injected cells. Total mortality was lower in patients treated with CD 34+ cells (14% vs. 35%; P=0.01) and the procedure was considered safe, with low morbimortality.
Several limitations of our study are recognized. First: the trial was projected with a small sample that was further reduced due to early mortality. This drawback was unexpected, since we had no early major complications or mortality in a previous safety study[11]. High mortality could be attributed to very low LVEF, which expressed extremely deteriorated LV function in those patients who died early. There was no low LVEF limitation for patient inclusion in the trial: all patients with LVEF<35% were candidates. Perhaps we could have avoided including patients with very low LVEF (for example, LVEF<25%) or, alternatively, once they were included, we could have employed special measures of support during and after treatment. Besides, since observed increments in contractility reported on several studies with cell therapy are small (in the range of 5 to 10%), the clinical impact of a small increase in heart function in a very ill population may be negligible. Due to these reasons, we can conclude that, in further studies, patients with LVEF<25% should be avoided. Secondly, we opted to analyze data by protocol instead of intention to treat, since patients who died had lower LVEF and maintaining their baseline data would have overestimated treatment effect. Thirdly, this was a non-blinded trial: it is possible that differences observed in quality of life and functional class could be due to placebo effect. Results from the 6-minute walk test would have been useful to assess more objectively functional capacity, but they were highly variable among patients, creating high values of standard deviation that precluded an appropriate analysis. Finally, high dispersion of data, from a wide range of parameters, such as LVEF and LV diameters, resulted in increased standard deviations in most outcome parameters, which in this small sample could have also precluded achievement of statistically significant differences between groups.
In conclusion, besides intragroup improvement in echocardiographic data, quality of life and NYHA class, when compared to control direct intramyocardial application of BMMC in NIDCM was not associated with sustained significant changes in left ventricular function. Functional capacity did not differ between groups. In future studies, less invasive forms of cell transplantation should be evaluated considering the high mortality observed, particularly for patients with severely impaired LVEF.
REFERENCES
1. Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, Ganiats TG, et al. 2009 focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation. Circulation. 2009;119(14):e391-479. [MedLine]
2. Manolio TA, Baughman KL, Rodeheffer R, Pearson TA, Bristow JD, Michels VV, et al. Prevalence and etiology of idiopathic dilated cardiomyopathy (summary of a National Heart, Lung, and Blood Institute workshop. Am J Cardiol. 1992;69(17):1458-66. [MedLine]
3. Kubo T, Matsumura Y, Kitaoka H, Okawa M, Hirota T, Hamada T, et al. Improvement in prognosis of dilated cardiomyopathy in the elderly over the past 20 years. J Cardiol. 2008;52(2):111-7. [MedLine]
4. Lloyd-Jones D, Adams R, Carnethon M, De Simone G, Ferguson TB, Flegal K, et al. Heart disease and stroke statistics-2009 update: a report from the American Heart Association statistics committee and stroke statistics subcommittee. Circulation. 2009;119(3):e21-181. [MedLine]
5. Seth S, Bhargava B, Narang R, Ray R, Mohanty S, Gulati G, et al. The ABCD Autologous Bone Marrow Cells in Dilated Cardiomyopathy) trial a long-term follow-up study. J Am Coll Cardiol. 2010;55(15):1643-4. [MedLine]
6. Fischer-Rasokat U, Assmus B, Seeger FH, Honold J, Leistner D, Fichtlscherer S, et al. A pilot trial to assess potential effects of selective intracoronary bone marrow-derived progenitor cell infusion in patients with nonischemic dilated cardiomyopathy: final 1-year results of the transplantation of progenitor cells and functional regeneration enhancement pilot trial in patients with nonischemic dilated cardiomyopathy. Circ Heart Fail. 2009;2(5):417-23. [MedLine]
7. Vrtovec B, Poglajen G, Lezaic L, Sever M, Domanovic D, Cernelc P, et al. Effects of intracoronary CD34+ stem cell transplantation in nonischemic dilated cardiomyopathy patients: 5-year follow-up. Circ Res. 2013;112(1):165-73. [MedLine]
8. Ishida M, Tomita S, Nakatani T, Fukuhara S, Hamamoto M, Nagaya N, et al. Bone marrow mononuclear cell transplantation had beneficial effects on doxorubicin-induced cardiomyopathy. J Heart Lung Transplant. 2004;23(4):436-45. [MedLine]
9. Nagaya N, Kangawa K, Itoh T, Iwase T, Murakami S, Miyahara Y, et al. Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation. 2005;112(8):1128-35. [MedLine]
10. Hofmann M, Wollert KC, Meyer GP, Menke A, Arseniev L, Hertenstein B, et al. Monitoring of bone marrow cell homing into the infarcted human myocardium. Circulation. 2005;111(17):2198-202. [MedLine]
11. Sant'anna RT, Kalil RA, Pretto Neto AS, Pivatto Júnior F, Fracasso J, Sant'anna JR, et al. Global contractility increment in nonischemic dilated cardiomyopathy after free wall-only intramyocardial injection of autologous bone marrow mononuclear cells: an insight over stem cells clinical mechanism of action. Cell Transplant. 2010;19(8):959-64. [MedLine]
12. Carvalho VO, Guimarães GV, Carrara D, Bacal F, Bocchi EA. Validation of the Portuguese version of the Minnesota Living with Heart Failure Questionnaire. Arq Bras Cardiol. 2009;93(1):39-44. [MedLine]
13. Levy WC, Mozaffarian D, Linker DT, Sutradhar SC, Anker SD, Cropp AB, et al. The Seattle Heart Failure Model: prediction of survival in heart failure. Circulation. 2006;113(11):1424-33. [MedLine]
14. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet. 1999;353(9169):2001-7. [MedLine]
15. Ly HQ, Nattel S. Stem cells are not proarrhythmic: letting the genie out of the bottle. Circulation. 2009;119(13):1824-31. [MedLine]
16. Macia E, Boyden PA. Stem cell therapy is proarrhythmic. Circulation. 2009;119(13):1814-23. [MedLine]
17. Abdel-Latif A, Bolli R, Tleyjeh IM, Montori VM, Perin EC, Hornung CA, et al. Adult bone marrow-derived cells for cardiac repair: a systematic review and meta-analysis. Arch Intern Med. 2007;167(10):989-97. [MedLine]
18. van Ramshorst J, Bax JJ, Beeres SL, Dibbets-Schneider P, Roes SD, Stokkel MP, et al. Intramyocardial bone marrow cell injection for chronic myocardial ischemia: a randomized controlled trial. JAMA 2009;301(19):1997-2004. [MedLine]
19. Zhao Q, Sun Y, Xia L, Chen A, Wang Z. Randomized study of mononuclear bone marrow cell transplantation in patients with coronary surgery. Ann Thorac Surg. 2008;86(6):1833-40. [MedLine]
20. Chang MG, Tung L, Sekar RB, Chang CY, Cysyk J, Dong P, et al. Proarrhythmic potential of mesenchymal stem cell transplantation revealed in an in vitro coculture model. Circulation. 2006;113(15):1832-41. [MedLine]
21. Beeres SL, Atsma DE, van der Laarse A, Pijnappels DA, van Tuyn J, Fibbe WE, et al. Human adult bone marrow mesenchymal stem cells repair experimental conduction block in rat cardiomyocyte cultures. J Am Coll Cardiol. 2005;46(10):1943-52. [MedLine]
22. Pijnappels DA, Schalij MJ, van Tuyn J, Ypey DL, de Vries AA, van der Wall EE, et al. Progressive increase in conduction velocity across human mesenchymal stem cells is mediated by enhanced electrical coupling. Cardiovasc Res. 2006;72(2):282-91. [MedLine]
23. Fukushima S, Varela-Carver A, Coppen SR, Yamahara K, Felkin LE, Lee J, et al. Direct intramyocardial but not intracoronary injection of bone marrow cells induces ventricular arrhythmias in a rat chronic ischemic heart failure model. Circulation. 2007;115(17):2254-61. [MedLine]
Financial support from Brazilian government agencies CNPq, CAPES, Ministry of Health and FAPERGS.
Authors' roles & responsibilities
RTSA: Analysis and/or interpretation of data, statistical analysis, final approval of the manuscript, conception and design of the study, realization of operations and/or experiments, drafting of the manuscript and critical review of the content
JF: Analysis and/or interpretation of data, realization of operations and/or experiments
FHV: Analysis and/or interpretation of data, statistical analysis, final approval of the manuscript conception and design of the study, realization of operations and/or experiments, Drafting of the manuscript and critical review of the content
IC: Analysis and/or interpretation of data, realization of operations and/or experiments
NBN: Analysis and/or interpretation of data, statistical analysis, final approval of the manuscript, realization of operations and/or experiments
JRMSA: Analysis and/or interpretation of data, realization of operations and/or experiments
IAN: Analysis and/or interpretation of data, final approval of the manuscript conception and design of the study, realization of operations and/or experiments
RAKK: Analysis and/or interpretation of data, statistical analysis, final approval of the manuscript conception and design of the study, realization of operations and/or experiments, drafting of the manuscript and critical review of the content
Article receive on Thursday, February 20, 2014
 All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license
All scientific articles published at www.bjcvs.org are licensed under a Creative Commons license